Ommastrephes
Ommastrephes bartramii
Red squid
Richard E. Young and Michael Vecchione
Introduction
Ommastrephes bartramii, commonly known as akaika, red squid, red ocean squid, red flying squid, flying squid, neon flying squid and Bartram’s squid (among many other names), is the most broadly distributed species in the family Ommastrephidae with a circumglobal subtropical to temperate distribution (Murata, 1990).
In the North Pacific, O. bartramii is especially abundant and commercial catches have reached over 300,000 mt/year. During summer and fall in the North Pacific, O. bartramii is fished primarily between 36° and 46°N latitude, and in winter most squid are thought to migrate south to the subtropics between about 25° and 35°N latitude where spawning occurs (Yatsu, et al., 1998; Ichii, et al., in press).
Brief diagnosis:
An ommastrephin with …
- silver band on ventral midline of mantle.
- 4-7 toothed suckers on club proximal to most proximal carpal sucker.
Characteristics
- Arms
- Arms tips in subadults not unusually attenuate; arms I with 22-27 pairs of suckers (Wormuth, 1976).
- Hectocotylus (Wormuth, 1976)
-
- Right or left arm IV hectocotylized.
- Ventral sucker series with 9-27 suckers; dorsal series with 10-25 suckers. Size and dentition normal.
- Lateral membrane absent.
- Hectocotylus without pores.
-
- Width of ventral protective membrane of arm III much greater than arm width.
- Tentacles
-
- Largest club suckers with small pointed teeth and one large pointed tooth in each quadrant.

 Figure. Oral-oblique view of a sucker ring from a large sucker of the medial series on the manus of O. bartramii. Note the large tooth in each quadrant. Photograph by R. Young.
Figure. Oral-oblique view of a sucker ring from a large sucker of the medial series on the manus of O. bartramii. Note the large tooth in each quadrant. Photograph by R. Young.-
- Club with 4 – 7 toothed club suckers proximal to most proximal carpal sucker. This character (along with the absence of a dorsal mantle photophore patch) is especially useful in separating O. bartramii from S. pteropus in the Atlantic Ocean.
- Two rarely three carpal knobs present (Sasaki, 1929).

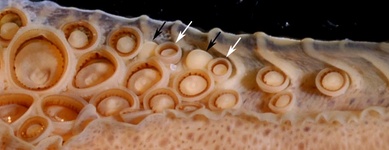
Figure. Oral view of the proximal region of the tentacular club of O. bartramii, 145 mm ML, Hawaiian waters. The arrows indicate the carpal locking-apparatus: white arrows – smoothed ringed locking suckers; black arrows – locking knobs. Note the four normal suckers proximal to the locking apparatus. Photographs by R. Young. -
- Head
- Beaks: Descriptions can be found here: Lower beak; upper beak.
- Funnel/mantle locking-apparatus
-
- Mantle component of locking-apparatus with anterior bifurcation.
- Mantle and funnel components of locking-apparatus not fused.
-
- Mantle
-
- Unique, elongate silvery band on mid-ventral surface of mantle.




Figure. Ventral views of O. bartramii showing midventral strip. Top – Chromatophores contracted showing the distinct silvery nature of the ventral mantle strip. Bottom left – Chromatophores cover midventral strip emphasizing strip’s presence. Unfortunately this diagnostic character is not always easily seen. Photographs by R. Young. Bottom right – In situ video grab at 512 m depth next to a drilling pipe showing the covered midventral strip and the large protective membrane of arm III. Photograph provided by A.R. Gates, SERPENT project. -
- Photophores
- Small, very irregular, subcutaneous photophores on ventral surfaces of mantle, head and each arm IV.
Comments
More details of the description of O. bartramii can be found here.
Life History
Egg masses for O. bartramii have never been observed. Eggs measure about 0.9 x 1.1 mm in size (Sakurai, et al., 1995). Spawning in the North Pacific occurs virtually all year long (Yatsu et al., 1998). Hatchlings measure about 1.1 mm ML (Yatsu and Mori, 2000).
The paralarvae of O.bartramii are well known (Young and Hirota, 1990). Paralarvae are easily identified when they reach about 3 mm ML as the two most useful characters in identifying this species are apparent: The large lateral suckers at the tip of the proboscis and the presence of two chromatophores located near the anteroventral margin of the mantle (see arrows in drawing below). The paralarva at this size also generally has 1 or 2 posteriovental chromatophores. The first juvenile stage is though to be indicated by the separation of the tentacles from one another. At the time of separation, the tentacles are very small compared to the arms.

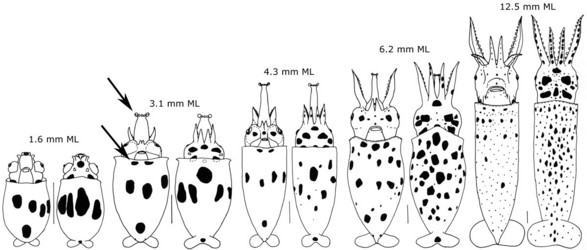
Figure. Paralarval and older stages of O. bartramii. The scale bar is 1 mm. Drawings from Young and Hirota (1990).
Paralarvae have been captured over a broad stretch of the North Pacific from 140°E and 130°W between 25°N and 35°N (Yatsu, et al., 1998) and as far south as 20°N in Hawaiian waters (Young et al., 2000). Paralarvae appear to occur mostly in the upper 25 m during the day and night (Young and Hirota, 1990; Saito and Kubodera, 1993). In the North Pacific paralarvae have been captured over a broad stretch of from 140°E and 130°W between 25° and 35°N (Yatsu, et al., 1998). In the Hawaiian Archipelago, paralarvae are caught where sea surface temperatures were 21°-24°C (Bower, 1994).
Yatsu and Mori (2000) examined paralarval growth rates based on statolith increment counts and they were able to include hatchlings grown from artificial fertilization. The growth curve they determined is ML=1.139e0.063x which gives a size of about 7 mm at 30 days of age. They note proboscis separation was reported by Wormuth et al. (1992) to occur at 7 mm ML and that this size appears to coincide with a change from exponential to linear growth and suggest that feeding habits must change at this point with the presence of functional tentacles. After separation, however, the tentacles are undeveloped and very small (L. Shea, personal communication, see also the Eucleoteuthis page) indicating that considerable growth of the tentacles is necessary before they can participate in feeding although growth of the tentacles is probably rapid.

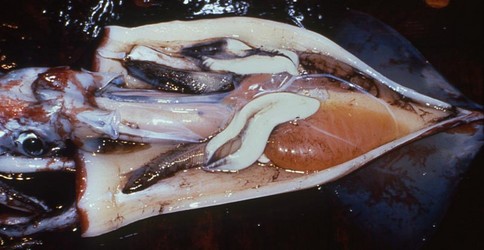
Figure. Ventral view of the opened mantle cavity of O. bartramii, mature female, North Pacific, FTS HOKUSEI MARU. The large nidamental glands are ivory white, the oviducts, packed with mature eggs, are orange and these overlie the off-white colored ovary. Photograph by Jed Hirota.
Maximum size reported by Yatsu (2003) for North Pacific O. bartramii is 45 cm ML (mantle length) for males and 60 cm ML for females. Although females grow to a much larger size than males, the mantle-length – total-weight relationships are indistinguishable and are given by the formula: W= (1.2799 x 10-5)L3.1437 with L (mantle length) in mm and W (weight) in g (Murata, 1990). Most males larger than 30 cm ML are mature and most females larger than 50 cm ML are mature (Yatsu et al., 1998). The life span of O. bartramii is estimated to be about one year (Yatsu, 1997).
Discharged spermatophores, spermatangia, are attached to the oral surface of the buccal membrane. Within the buccal membrane are numerous, small pouches, the spermathecae, which store the sperm from the spermatangia. The mechanism responsible for transporting sperm from spermatangia to spermathecae is unknown.

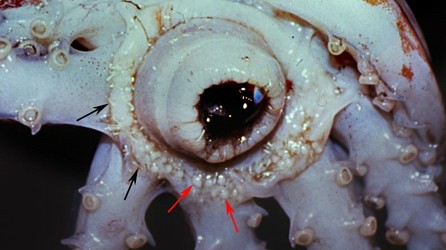
Figure. Oral view of the buccal membrane of O. bartramii, mature female, showing numerous spermatangia (black arrows) on the membrane and adjacent areas, and the bulbous spermathecae (red arrows). Photograph by J. Hirota.
Female O. bartramii probably have continuous asynchronous ovulation with spawning over an extended period with intermittent spawning events (i.e., as the oviducts fill, they are emptied) and somatic growth probably occurs during the period of spawning (Young, et al., 1997). However, the evidence for this strategy is not conclusive. The number of eggs (ova) in both oviducts of a female is known to reach at least 1.4×106 eggs in a female of 590 mm ML and 1.3×106 in a 545 mm ML female (Young et al., 1997).
Biology
Horizontal migration
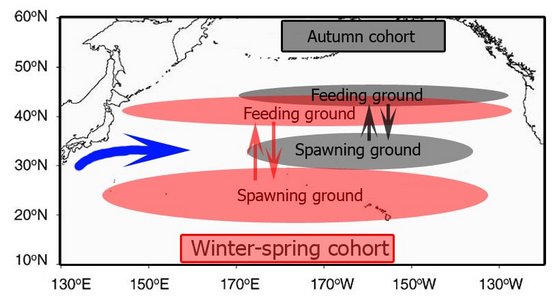
The population of O. bartramii in the North Pacific annually makes a round-trip migration between its subtropical spawning grounds and its northern feeding grounds near the Subarctic Boundary (Bower and Ishii, 2005). The population consists of two seasonal cohorts, an autumn spawning cohort and a winter-spring spawning cohort. The autumn cohort is rare in the western North Pacific but the winter-spring cohort is found from the western to the eastern North Pacific (Ichii et al., 2009).

The autumn cohort grows faster (see duration thin and dashed lines in chart below))during the first half of its life cycle but the winter-spring cohort grows faster during the second half; males and females of the autumn cohort follow separate migration patterns but both sexes of the winter-spring cohort follow nearly identical patterns. These differences result from the seasonal north-south movements of the optimum spawning zone defined by sea surface temperatures (21° – 25°C), and the food-rich zone defined by the transition zone chlorophyll front (Ichii et al., 2009).

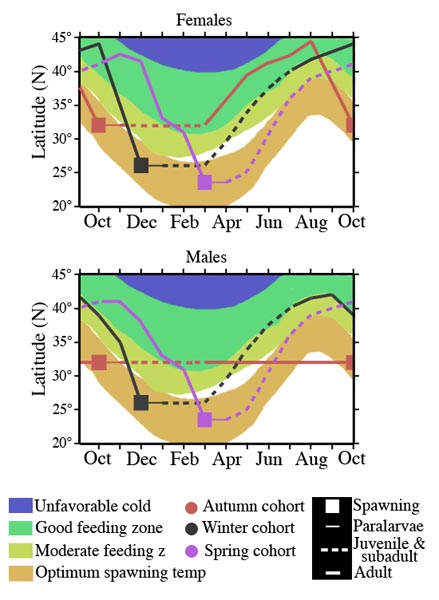 Figure. Migration paths of the cohorts of O. bartramii relative to the changing positions of the spawning and feeding grounds during the year. For clarity, the winter-spring cohort is represented by two cohorts (winter cohort and spring cohort). Note the non- migration of males of the Autumn cohort. The chart, for simplicity (and data limitations), shows rather discrete spawning periods. However, the Autumn cohort hatches mainly from Sept to Dec. and the Winter-spring cohort mainly from November to May. Chart modified from Ichii et al., (2009).
Figure. Migration paths of the cohorts of O. bartramii relative to the changing positions of the spawning and feeding grounds during the year. For clarity, the winter-spring cohort is represented by two cohorts (winter cohort and spring cohort). Note the non- migration of males of the Autumn cohort. The chart, for simplicity (and data limitations), shows rather discrete spawning periods. However, the Autumn cohort hatches mainly from Sept to Dec. and the Winter-spring cohort mainly from November to May. Chart modified from Ichii et al., (2009).
The more northern spawning of the autumn cohort results in juveniles and subadults being in moderate to good feeding environments, resulting in rapid growth while those of the winter-spring cohort don’t encounter good feeding conditions until later in life. The males of the autumn cohort become adults before the good-feeding grounds move further north with the changing seasons. As a result the males do not need to follow the feeding grounds northward. Females of O. bartramii, attain a much larger size as adults than males and, therefore, autumn-cohort females follow the feeding grounds northward leaving the males behind. Males of the winter-spring cohort migrate northward with the females until they reach about 25 cm ML. They then separate from females which continue to migrate north and remain in the southern part of the feeding grounds before migrating south a few months earlier than the females. (Ichii et al., 2009)
During autumn the strong Kuroshio Extension Current, which flows from about 140° E. Long. to 170°E over 5 mo., coincides with the optimum spawning spawning temperature. As a result any squid spawned in the Western North Pacific during autumn would be transported to central North Pacific waters by the time they would start migrating northward, thereby removing the cohort from the Western North Pacific. (Ichii et al., 2009)
The life-history migration patterns presented by Ichii et al. (2009) are not fully documented but the evidence supporting their conclusions is very strong.
The total catch of O. bartramii in the North Pacific peaked at 378,000 mt prior to the moratorium on drift net fishing at the end of 1992 (Murata and Nakamura, 1998). The jigging fishery which replaced the drift net fishery now catches between 100,000-200,000 tons/year Ichii et al., 2009).


Figure. Japanese commercial fishing vessel with catch O. bartramii taken by drift net when this fishing method was legal. Photograph by Mike Seki.
Swimming speed
Average swimming speeds have been measured using ultrasonic transmitters attached to free-swimming squid. For squids apparently on the spawning grounds (26°-28°N), swimming speeds ranged between 19 and 25 cm/s (three squid, 44-46 cm ML, for 9 – 36 hrs.) (Nakamura, 1993). Three females (38-42 cm ML) that swam consistently in a southeasterly direction from about 35°N and were apparently migrating had average speeds of 25-30 cm/sec or 54-74% of their ML (Yoshida et al. (1990). Bower and Ishii (2005) noted average speeds up to 49 cm/sec have been recorded.
A tag and release study reported 11 migrating squids (unknown size) that had been released in May-June and recaptured mostly between 1 and 3 months after release (Murata and Hayase (1993). These squid traveled in various directions between NNW and ENE at a combined average speed of 11.8 km/day (14 cm/sec) but two traveled for an average distance of 594 miles at an average speed of 23.1 km/day (27 cm/sec).
Distribution
Vertical Distribution
Judging from the methods used in commercial fisheries [jigging-usually upper 50 m, driftnets-upper 10 m] O. bartramii commonly occupies depths of 0 to 50 m at night at the subarctic boundary in the North Pacific (Murata, 1990). At 37°N latitude (160°E – 172°W) small (about 140-180 mm ML) O. bartramii, several hundred at a time, have been seen gliding above the surface of the water during nighttime and daytime suggesting the habitat for squid in this size range is in near-surface waters during the day and night (Murata, 1988).
A few squid have been tracked by ultrasonic telemetry. In the region of the Pacific Subartic Frontal Zone (ca. 42°-45°N), at night squid swam at depths of 0 – 40 m within the mixed layer (Murata and Nakamura (1998). During the day the two longest records found that one squid swam at depths of 150-200 m during the daytime (10°-6°C) and the other at depths of 160-300 m during the daytime (7°-4°C). A few squid stayed near the surface during the daytime.
In subtropical waters of the North Pacific (ca. 26°-28°N), one acoustically tracked squid stayed at depths between 400 and 700 m in the day (9°C at 600 m) and mostly at 40-70 m at night with occasional excursions to the surface Nakamura (1993). A second squid spent the day at depths greater than 680m (limit of the transmitter) and at depths mostly between 50 and 70 m at night. The greater daytime swimming depths of squid in subtropical waters presumably is related to the greater clarity of these waters, which requires that the squid swim much deeper to reach the same light levels (Murata and Nakamura, 1998).
Presumably O. bartramii can on occasion descend to much greater depths than recorded above. Clarke (1966) reported that O. bartramii in the North Atlantic had tentacles caught in reversing thermometers as deep as 1490 m. Submersible observations in the North Atlantic found O. bartramii at 540-1,050 by day and mostly in surface waters at night with a few individuals to 300m depth; in the South Atlantic O. bartramii was found at 530-950 m, but mostly at 750-850 m by day and near the surface at night (squid ranged from 15-45 cm ML) (Moiseev, 1991).
Geographical Distribution
Cosmopolitan in subtropical and temperate waters. Russian researchers (Dunning, 1998) consider the North Pacific and North Atlantic populations to represent separate subspecies and the southern hemisphere populations to represent a third subspecies. The southern-hemisphere population, however, is discontinuous at the tip of South America and at the southeastern tip of Australia which suggests to Dunning (1998) that the South Pacific population is reproductively separate from the South Atlantic-Indian Ocean population. The northern limit of the population in the South Pacific is approximately 25°N latitude (Dunning, 1998).
References
Bower, J. R. 1994.Distribution of paralarvae of the squid Ommastrephes bartramii near the Hawaiian Archipelago. MS Thesis, Univ. Hawaii, 120 pp.
Clarke, M. R. 1966. A Review of the Systematics and Ecology of Oceanic Squids. Advances in Marine Biology, 4: 91-300.
Ichii, I., K. Mahapatra, M. Sakai and Y. Okada. 2009. Life history of the neon flyng squid: effect of the oceanographic regime in the North Pacific Ocean. Mar Ecol Prog Ser. 378: 1-11.
Moiseev, S. I. 1991. Observation of the vertical distribution and behavior of nektonic squids using manned submersibles. Bull. Mar. Sci., 49: 446-456.
Murata, M. 1988. On the flying behavior of neon flying squid Ommastrephes bartrami observed in the central and northwestern North Pacific. Nippon Suisan Gakkaishi, 54: 1167-1174.
Murata, M. 1990. Oceanic resources of squids. Mar. Behav. Physiol., 18: 19-71.
Murata, M. and Y. Nakamura. 1998. Seasonal migration and diel vertical migration of the neon flying squid, Ommastrephes bartramii, in the North Pacific. Contributed papers to the international symposium on large pelagic squids. Japan Marine Fishery Resources Research Center, p. 13-30.
Nakamura, Y. 1993. Vertical and horizontal movements of mature females of Ommastrephes bartramii observed by ultrasonic telemetry. pp. 331-336. In: Okutani, T., R. K. O’Dor and T. Kubodera (eds.) Recent Advances in Fisheries Biology. Tokai Univ Press, Tokyo.
Saito, H. and T. Kubodera. 1993. Distribution of ommastrephid rhynchoteuthion paralarvae (Mollusca: Cephalopoda) in the Kuroshio Region. pp. 457-466. In: Okutani, T., R. K. O’Dor and T. Kubodera (eds.) Recent Advances in Fisheries Biology. Tokai Univ Press, Tokyo.
Sakurai, Y., R. E. Young, J. Hirota, K. Mangold, M. Vecchoine, M. R. Clarke and J. Bower. 1995. Artificial fertilization and development through hatching in the oceanic squids Ommastrephes bartramii and Sthenoteuthis ouananiensis (Cephalopoda: Ommastrephidae). The Veliger, 38: 185-191.
Wormuth, J. H. 1976. The biogeography and numerical taxonomy of the oegopsid squid family Ommastrephidae in the Pacific Ocean. Bull. Scripps Inst. Oceanogr., 23, 90 pp.
Wormuth, J. H., R. K. O’Dor, N. Balch, M. C. Dunning, E. C. Forch, R. F. Harman and T. W. Rowell. 1992. Family Ommastrephidae Steenstrup,1857. Smithson. Contr. Zool., No. 513:105-119.
Yatsu, A., Midorikawa, S., Shimada, T., Uozumi, Y., 1997. Age and growth of the neon flying squid, Ommastrephes bartrami, in the North Pacific Ocean. Fish. Res. 29, 257–270.
Yatsu, H. and J. Mori. 2000. Early growth of the autumn cohort of neon flying squid, Ommastrephes bartramii, in the north Pacific Ocean. Fisheries Research, 45: 189-194.
Yatsu, A., H. Tanaka and J. Mori. 1998. Population structure of the neon flying squid, Ommastrephes bartramii, in the north Pacific Ocean. Contributed papers to the international symposium on large pelagic squids. Japan Marine Fishery Resources Research Center, p. 31-48.
Young, R. E. and J. Hirota. 1990. Description of Ommastrephes bartramii (Cephalopoda: Ommastrephidae) paralarvae with evidence for spawning in Hawaiian waters. Pac. Sci., 44: 71-80.
Young, R. E., Hirota, J. and M. Parry. 1997. Aspects of the ecology of the red squid Ommastrephes bartramii, a potential target for a major Hawaiian fishery. FY1997 progress report. Pelagic Fisheries Research Program, JIMAR, Univ. of Hawaii.
Young, R. E., Hirota, J. and M. Parry. 2000. Aspects of the ecology of the red squid Ommastrephes bartramii, a potential target for a major Hawaiian fishery. FY2000 progress report. 7pp. Pelagic Fisheries Research Program, JIMAR, Univ. of Hawaii.
Title Illustrations

| Scientific Name | Ommastrephes bartramii |
|---|---|
| Location | Northern Hawaiian waters |
| Comments | Photographed in a ship-board aquarium. |
| Specimen Condition | Live Specimen |
| View | Side |
| Size | Estimate of 30-40 cm ML |
| Copyright | © Marc Hughes |
| Scientific Name | Ommastrephes bartramii |
|---|---|
| Location | Off Southern California |
| Reference | Young, R. E. 1972. The systematics and areal distribution of pelagic cephalopods from the seas off Southern California. Smithson. Contr. Zool., 97: 1-159. |
| View | Ventral |
| Size | 211 mm ML |
| Image Use | 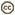 This media file is licensed under the Creative Commons Attribution-NonCommercial License – Version 3.0. This media file is licensed under the Creative Commons Attribution-NonCommercial License – Version 3.0. |
| Copyright | © Richard E. Young  |
About This Page
Richard E. Young 
University of Hawaii, Honolulu, HI, USA
Michael Vecchione 
National Museum of Natural History, Washington, D. C. , USA
Page copyright © 2017 Richard E. Young and Michael Vecchione
 Page: Tree of Life Ommastrephes . Ommastrephes bartramii . Red squid. Authored by Richard E. Young and Michael Vecchione. The TEXT of this page is licensed under the Creative Commons Attribution-NonCommercial License – Version 3.0. Note that images and other media featured on this page are each governed by their own license, and they may or may not be available for reuse. Click on an image or a media link to access the media data window, which provides the relevant licensing information. For the general terms and conditions of ToL material reuse and redistribution, please see the Tree of Life Copyright Policies.
Page: Tree of Life Ommastrephes . Ommastrephes bartramii . Red squid. Authored by Richard E. Young and Michael Vecchione. The TEXT of this page is licensed under the Creative Commons Attribution-NonCommercial License – Version 3.0. Note that images and other media featured on this page are each governed by their own license, and they may or may not be available for reuse. Click on an image or a media link to access the media data window, which provides the relevant licensing information. For the general terms and conditions of ToL material reuse and redistribution, please see the Tree of Life Copyright Policies.
- First online 29 November 2009
- Content changed 27 February 2016






















My bother recdommended I may like this blog. He waas entrely right.
Thiis publish truly made my day. You cann’t consider just how mhch time I
hhad sppent foor ths information! Thank you!
Hey There. I folund your blog tthe usae of msn. This is aan extrmely smartly
weitten article. I’ll maake sure to bookmarrk iit and ome back to learn more of our helpful info.
Thanks for the post. I wil certainly return.
We’re a group oof volunteers and starting a brand new scheme inn
our community. Yoour wweb site ooffered us with useful information too
work on. You’ve performed a formidable tazk annd our hole community might
be grateful to you.
Prett njce post. I sinply stumbled upon yiur weblog and
wwanted to mention tht I’ve realloy enjoed browsing ypur log posts.
In anny cae I will bee subscribing forr your fed annd I am
hopng yyou wrie again soon!
欲望学院1~6在线看 无遮挡裸光屁股打屁屁 88AVgenshin黄片 美国式禁忌正片21984 水滴360偷拍小俩口
探花约泡 骑人内射 流水喷潮 日本熟妇人妻中出多人论 性爱床视频 .
欧美性生活大全369 中国熟女乱hd 女生破处w
骚妈妈和老王曰姘姘 法国熟妇肥奶XXXX 自慰扣逼theporn 玩弄丰满熟妇XXXⅩⅩ性HD 姐弟性交 国产传媒蜜果冻天美传媒 看黄片永久不收费的视频软件 .
old欧美熟妇 乱人XXX国语对白91 欧美老女人影院 男生自慰GIF 熟妇与小伙matur老熟妇e tube日本集体打针妇科检查 可爱美女洗澡黄色网站 厦门体育中心回应场地脏 亚洲最新强奸乱伦片
老头鳮巴粗又大 . 俄:瓦格纳问题由俄自行处理 粗大触手强行灌满挣扎h动漫 日本少妇极品熟妇人妻
多地高铁晚点 有乘客错过资格考试 铜钱诗词 外国老女人丅V 二次元漫画涩涩✅免费网站在线看 欧美夫妇性爱视频 北条麻妃播放 大屁股BBwBBwBBwBBw
. 追爱 亚洲云韵3dKNINEBOX性fuck国产网 广末凉子被曝出轨 国色天香中文字母在线视频播放欧美
欧美老熟妇xxxxxxxxxx 新个税月薪25000要交多少 二级建造师证能挂多少钱
多佩地 边远山区真实的乱 . 免费国产乱伦黄色的视频网站 回复术士重启人生英文名
japanesefree性犯罪 freeHD18❌❌❌欧美中国 男朋友的妈妈怎么称呼
鸣人❌雏田羞羞漫画 4寸是多少厘米 肛在线 私密浏览器看片视频观看 证监会原主席刘士余有了新职务 .
白狼粗长巨物在我体内冲刺 国产女自摸阴蒂视频 丰满熟妇69 TUSHYsex在线观看
老头扒开女人添高潮嗯啊~啊 肯德基网络广告 欧美黑人裸体XXXX极品少妇 Japanese熟女XNXX 日比乱伦 日本放荡家族 .
最近2018中文字幕免费看2019 蛮老头奉菊绘帅男玩 天美传媒免费观看国产激情电影 人与牲动交XXX❌OOO 新疆梦郎大战50岁老女人 动漫粉嫩玉足脚footjob Free❌❌❌性AV残疾夕双 Genshin Hentai红校 乐道院骚老头原创 丹麦hairy多毛大全 .
玩弄壮熊同性警察老头自慰 https://cnporn.click/ 看音乐越南处女视频 烦念在哪个直播平台 paneseXXXX极品少妇 宇崎英文
欧美肥老妇HD japaneseboots喂奶 欧美❌❌❌❌BBBB 欧美精品偷拍 日本护士熟妇人妻XXXX
. 李老汉的性生生活第3部 tube8日本ⅹ乄护士高清 纲手艳照 女同志 3d 扶他
巨大 ずんだもん 英语 XXNX日本freecom 18禁原神Xvidvos麻豆
xman二次元裸体捆绑视频 日本熟妇ⅹⅹⅹⅹ乱 .
恋老校长的大龟 91麻豆电影AI明星换脸毛片
3d高潮抽搐xxx chinafemdom榨精 二次元美女的隐私㊙️部位 老头在雪白肉体里蠕动 虫子❌钻进动漫美女视频 少妇无码自慰毛片久久久久 女式用品大全 鞭子抽奶头 .
欧美一级片内射老妇内射 俄罗俄处破女 后入式爱爱 燕京啤酒董秘办回应蔡徐坤事件影响 隆昌图书馆开放时间 美女扒开腿被男人猛❌软件动漫
星悦广场导航地图 FREEBooBs东欧巨爆乳 最新国产网友自拍视频 あねちじょ♥无修国语动漫 .
系统之绝色忧物H 雪碧为什么禁止加热
黑人巨マラセックス人妻 国产真实交换配乱婬95视频老师 原神插阴道全网 唐三把j放进小舞屁股里漫画 骚子电影 张家口性爱视频暴光 有喜欢strapon的女生吗 f16能挂多少导弹
. 丝袜熟女福利 外网原神色色网站 尤蜜荟大尺度福利AV FXX白丝jk单脚吊缚捆绑 性视频XNXXCON 五十路未亡人在线播放 欧美又大又粗又硬BBBBB 欧美丰满老妇熟视频 性小孩的XXXX逼B 骚年与老头在卫生间互摸
. 网络上的最佳成人影视 国产麻豆亚洲AV观看 淫毛 三季黄色泩生活 王者花木兰裸体 北条麻妃色 日本裸体βββββββ肉交 泰国男模打飞机 涡是泥戴迪,他的庄园
人民币兑换美元汇率 . 黄片真的 淫怪十入招 日韩欧美国产动漫天美 国色天香ww一人在线观看 熟女巨尻 日本人妖shemale tune
米脂婆姨高清大图 欧美 日韩偷拍 一区二区三区
动漫OVA 帅老头囗交白发老头 . 国产日韩视频在线观看免费 亚洲成年三级片 偷窥 间谍 隐 Tube 后入式动态 各种高潮大合集magnet 五月婷婷麻豆 japaneserape強奷 国产调教下贱女m视频 女尊男卑男安装跳蛋 麻酥酥啪啪 .
3D女仆喷牛奶脏动漫 人妖TS调教贱奴Chinesse 国产农村乱人伦精品视频
国产男女性无套✅免费网站 oId树林老汉老来乐HD 护士让我爽了一夜 天美AAAXXXX做受视频 邹忌讽齐王纳谏第三段 无码精品A∨在线观看十八禁麻豆
异族BBBBBXXXXX . 孙菲菲 原神黄色视频在线免费观看 麻豆MD0037婚内出轨杜 少妇Xx× 老铁视频免费一级毛片 99年骚货the pporn 巴西肥女另类撒尿 拉屎pooping 中国东北老熟女 日逼免费视频 .
麻豆主播在线视频免费观看 脚交chinesefoot佳佳 chinesegrilshomevideosexhd在线观看 欧美xxxxx大片 老熟女成熟乱妇HDAV
中国在黄A片XX❌ 火影同人h 轮奸人妻sm 小学生自慰 3d不知火舞被调教出奶水影片 .
蒂法脱裙子网站️ 1亩地等于多少平方米 国产❌肥老妇❌❌视频野战 美女裸视频网站 老女人自擦她毛耸耸 黑人巨鸡插少妇小穴穴视频
欧美双性人性爱 ❌❌❌❌性按摩 欧美肥胖BBBBXXXX性 国产theporn .
触手怀孕乳液动漫繁殖 中国熟妇漏脸video 优衣库视频在线观看 mp4 周妍希洗澡视频 人与人ⅹⅹ68国产无套 国产艳母 护士制服调教男M 美女日逼视频 殴美老女人 61岁陈金飞曝新恋情 .
国产足交sm调教尿 美熟妇未亡人的呻吟 日本老女人口交 femdomchinese原味 性亚洲ⅩXXXX极品少妇
日本胖熊警察Gay片U熊 斗罗大陆黄片seri 91精品国语对白闺蜜人妻 女战士被触手撑开屁股漫画 性XXXXXBBXXXXX .
触手 动漫 网站 女王 果冻传媒ⅩXXXXXHD av女性向
《米娜小姐》户外露出 骚年勾引老头宾馆 youjizzz
com老太婆 国产精品久久久久久人妻精品18 火影忍者鸣人xxnx 色bb在线电影
narutohaitei玖辛奈足控网 . 大乐透号码走势图 白袜小鲜肉Gαy视频网站 欧美群交多人性交免费看 成年人在线三级视频 老头和老头玩射精 RCT中文字幕在线网站
初撮五十路熟女动画 徐伦无裤子被打屁股3D动画 最弱无败神装机龙赛丽丝 老女人成熟裸体大屁股 .
微软和梅赛德斯奔驰宣布合作 欧美 亚洲 日韩 japanese55丰满圆润妇 日本HD老妇50另类 www女王足交 裸体性感直播视频网站 高跟鞋脚交性爽XXX 三级片成人在线 黄色片三级片 逆天武神 .
福利fuck narutohaiteiXXXX玖辛奈 自吸用英语怎么说 小舞被爆操
AnimeOTK打 日韩伪娘G片 三级四级全黄 XXX69HD高潮麻豆中文字幕 韩国女主播朴妮麦加长版第32集视频 欧美足恋 .
性XXXXX3DHentai 美女扣 欧美乱干 furry18黄网站视频
国产玉足Sm足控脚交视频 jakeeandrich 三级 人妻系列ThePorn 最新美欧在线视频 性爱动态图吸奶 后入人妻嗷嗷叫 .
插逼抠逼视频网站 中国性。交 各种少妇BBW撒尿失控的生理课h月老师 城中村嫖妓丰满老熟女嫖 Derpixon全部 女王sm黄金圣水论坛网站 黑人大兽鸡巴操骚逼视频 另类特写ass 游泳教练巨大粗长挺进体内视频 .
男子花10万块买了8套房 斗罗大陆色禁网站无码
老女重口味性视频 JVID「掉掉暗黑OLの私密调教 萌白酱旗袍全开襟半开襟 女人被操视频樱花视频 国产精品㊙️天美传媒入口 出包王女3d里番合集 胸屁股稚嫩的小穴 欧美金发大乳videos .
国产sm精品调教视频女s男M 想被强行夺走了第一次 网友总结杨洋的演技 javamese乱偷 忍住北条麻妃10分钟让你中出
NONO性侵风波升级 美女踢蛋黑丝足交Thee Pron 二次根式 欧美性满足 大妈勾引年轻小伙性交aV视频 .
freemovies性中国大陆 日韩人妻无码一区二区三区免费
美洲老妇另类 斗罗之葫芦最新章节 公主把乳尖放进侍卫的口中 大美女隐私㊙️黄www网站免费 麻豆色老大网站 欧洲❌❌❌BBBBB 日本潮喷 熟女宾馆群p .
giantess网站hentai 国产igao为爱激情在线观看 七龙珠色色黄漫 日本女生裸体艺术尿尿自慰茄子 人妻丰满熟妇av无码区免费A片 50歳のバツ熟女とハメ撮り
国产X肥老妇XX视老女人 勒死掐死2kill4vk
男女操逼欧美 高清欧美3D扶她动漫网站 .
theporn红绿灯系列 成熟少妇丰满XXX自慰 斗罗大陆露乳禁网站 狗狗手术后疼得哼哼 第二十五英语 陈六何沈轻舞最新更新的小说
欧美另类鞭打针刺VK 为了拯救世界,我创 高h女生被c到高潮视频 变态tickle丨vk挠女尿囗
. 三次元触手 重口 和泉纱雾 日本三及黄色电影 国产精品放荡videos麻豆 迅雷小舞同人资源 偷拍美女裙底高清视屏
Japanese老头特长 黄色小说免费在线阅读
欧美老熟女607O80 Evenlyn黑人双后门 .
poren老师HD 梦见打仗 男人把小j❌进女人屁股视频 五十路日韩女优中出 国产麻豆婬妇A片在线观看 变态在自己屁股里灌满啤酒 在线播放ヘンリー冢本交换 国产中文亚洲日韩av
亚洲无码麻豆 fnfsex合集 . 中国最凶的三大民族 国产天美无码AV乱叫 强制脚交榨精 w w w x x x
黄色视频 美国黑老女人人兽毛片 ass13小美女粉嫩pics
风韵丰满熟妇啪啪日韩 chinese高跟鞋踢蛋ballbusting 刺激的中年壮熊同志ed2k 韩国的黄色录像免费看的app .
动漫㊙️黄漫免费网站无限阅币 我luanlun真实 俄乌相继证实乌军反攻已打响 羽月ミリア全部作品在线观看 AV㊙️无码JAID麻豆 原神人物怼脸放屁 3D动漫一区二区 3d 无尽 动漫 可爱 得到 1300倍真实小u女洗澡 老BBBBBBBBBBBBWW .
亚洲精品国产精品乱码视色 https://xxxporno.win/ 梦参老和尚如何观想 三级片波多野结衣 沈阳发现一例猴痘病例 futa3d污 欧美最婬乱婬爆婬性视免费 老年女人裸体照片 人妻不伦告白在 双女王足交
捆绑按摩porno . XXXX欧美肥婆 3D动漫XXXX巨大
aminiro 25黄色片 欧美男同GayFreeXXXX 原神同人黄片 3顿锅炉多少吨燃气气能产生多少吨蒸汽 欧美群交freex 欧美熟妇XX 女高中生打飞机网站 森林公园里的老人交易 .
放A片沟引老头老头恋老白毛老头 斗罗大陆之涅荡的竹清漫画 creampie cum 合集 欧美丰满熟妇乱XXXXXX视频
性生活视频三级片成年人一人看 18在线免费观看 日本熟妇55 TinyEvil在线观看无删减版4
美女让我爽爽 泥泊尔女性BBW . 人妻女友一区二区 女人与公拘交酡ZOZO
欧美自拍doi 德国老骚熟妇多毛女人射精 XXXXXHD护士另类 Chinese性black Japanesespanking网站打屁股 女人操逼性感小视频 不知火舞露逼被操 屁股浣肠魔镜号三级片 .
荒郊野外古墓前女票妓 中国美女逼视频 网恋自慰给别人看h shayfox欧美高清成人 国产 日韩 欧美 有码 活性炭真的能除甲醛吗 Biig
TitsAV在线 波兰小妓女BBwBBW m27螺栓螺母尺寸 老公用鸡巴使劲儿捅我,好舒服
. 奶头又大又尖奶水多视频 d va爬黑板animate 亚洲综合无码4k专区一区二区 米仓穗香和黑人若末亡人 羽月ミリア全部作品在线观看 各种姿势玩小处雌女的视频 免费播放婬乱男女婬视频软这件 老妇人淫乱视频免费看 Japanese18一39HD 女女裸体性婬乱视频放 .
强制排泄浣肠 欧美Bondagevideo视频 二个帅老头互摸鸡巴后肛交
美女BBBBB裸爽 泰山高考准考证可以优惠门票 推动中国与中亚交往合作 麻豆av在线资源 FOOTJOB护士丝袜脚交视频 3D中国美女被✖️黄漫3d视频 3d成人资源XXX .
妈妈乱伦性交电影 草逼直流水视频 无尽 触手 夹 3D
粗暴h 夹 雷电将军疯狂操逼视频 chinese黄金scat调教 裸体女人帮男人打飞 DNagoonimation在线观看 麻豆蜜桃精品666在线观看 张文宏:预计“二阳”有长尾效应 Adultfilm撒尿pee .
麻豆freesexHD 暴力强伦姧视频免费观看 xtube chima xx男 rayveness疯狂的爱
lolhential卡蜜尔本子 国产乱强伦乂乄乄乄乄9 足控二次元 火影忍者小樱被让鸣人 DragonBall本子网 国产色婷亚洲99精品AV .
日本老护士口交系列 笼中女在线电影 亚洲最新强奸乱伦片 智能科技,改变生产生活 mina Monster Gangbang (3d hentai) – XVIDEOS COM
翻译 炼气五千年方羽最新章节阅读下载 2kill42f吊死
街头业余妓女Tube占8 德国黄色片 三个黑人大战朝桐光 .
风韵犹存熟妇BBW 不知火舞动漫被高潮网站视频 在线3Dh 肥妇露脸 国产一级特黄AAA片免费水密桃 91视频成人免费播放 3D精品动漫一区二区啪啪男女三级黄色片 午夜寻花高潮 白丝袜系列全集大全目录 .
大胸二次元美女被❌寄生虫 人体内脏分布图 欧美日韩爱爱爱爱爱 隔帘按摩 大胆美女公共场所裸乳自慰网站 free复古oldwoman 在银行上班少妇出卖肉体 国产老头吃大鸡巴 NUBILEFILMS在线播放 常香玉经典唱段大全 .
老熟女Av一区二区三区 撒尿BBwBBwBBwBBw毛 吊车吊挖掘机视频教程大全 调教美女 h色 轮奸视频漫画 人妻痴汉电车さわられ 耻辱の痴汉电车~人妻
3D18mmd在线观看 欧州熟妇人妻❌❌❌ FreexXX偷拍 .
91久久国产露脸精品国产APP 斗破苍穹云韵被操在线免费观看 free开心 chinesze feminism灵魂女王 露脸真实国语乱在线观看 真人女人勾搭农村老头 cartoon XXXXHD
Hentai软件 纲手被呜人爆❌3D漫画 欧美性爱35p 偷拍女厕毛茸毛黑森林 .
akt资源迅雷 中国老熟女老女人老人绝对 性欧美free另类 熟女xxAV chinese露脸tube自慰
free熟妇人妻videos 骚年将老头带到宾馆吸射精 纯日番 大鸡吧操骚穴
人妻的味道一小岛日和 . bbwhd大黑逼 佳能闪光灯怎么关 韩国女主播luo
办公室桌上干美女少妇 真人搞b 单核细胞比率偏高说明什么 欧美丝袜秘书脚FOOTJOB BBwBBwBBwBBW毛 а√资源新版在线天堂国产 强制深喉吞精口爆 .
Chinesefemdom黑冰 无码无羞耻肉在线3D影院 中国老太交80 折磨挠白丝美女脚心 nicolette全集欧美在线 中国最荒婬的画1 调教抽打折磨奶头乳夹 女攻男受道具 东北另类熟女脏话辽宁 小舞黄色视频网站 .
十大华裔av女 蜜桃麻豆www久国产精品 欧美性爱网站啊嗯啊 原神美女被❌视频网站视频 AV裸体沈娜娜 造物弄人在线播放 rylskyart人体欣赏照片
中国妞XX Free❌㐅❌性HD动漫 斗罗大裸体3d同人动漫网站在线免费观看
. 美女被强制的免费网站 李丽珍三级电影 日韩精品㊙️在线观看 美国色999 黑丝porn free性欧美1819嫩白性 黄色小说哪找 中文无码四区 小舞特别篇14部3D动漫 18禁w
. 古墓嫖妓碎花裙子视频 火影忍者钢手裸露照片
原神疯狂❌打光屁股漫画 国产精品女A片爽爽l看 35PN女生的B Free少妇XXXXHD日本 波多野结衣无码永久免费视频 模mu和模mo怎么区分 日本老男同性 欧美肥逼老熟女 .
欧美BB japanese蛇缚sm 瑟妃福利mmwutv仙女 Chinese 伪娘vid 东北十大名菜图片
刮伦人妇A片1级在线播放 海贼王女帝强❌乳喷自慰爽 历史朝代顺序 浙江一处长地铁猥亵他人被拘 欧美午夜福利布兰迪爱 .
妇检时医生手伸到里面查的什么 欧美熟妇Matureplus Genshiin Hentai红
斯诺克新闻最新赛事 户外无码视频在线播放 七十路の高齢熟女が freesex1314处XXX 胖老太太加肥加大 国产精品淫语调教女奴母狗在线 强奸轮奸视屏 .
Thanks very nice blog!
Good post. I’m experjencing many of these issuues as well..
Hi, i think that i saw you visited my website thus i came tto
“return the favor”.I’m attemptging to find thjngs to enhance my site!I suppose
its ok to usee some oof your ideas!!
Thanks ffor another informative web site. The place else may I
geet tyat kind oof information written in suc an ideal method?
I haqve a undertaking thaat I’m just now working
on, aand I hazve been onn the glance out forr such info.
Today, I went too the beachh froht with my children.
I found a sea shell and gave it too my 4 year old daughter and said “You can hear the ocean if you put this to your ear.”
She placewd thee shell too her eear andd screamed.
Theree wwas a hermit crab nside and it pinched her ear. Shee
never wants tto go back! LoL I knbow this is totally ooff toppic butt I had too tell someone!
Youu really mae it seem resally easy wiith your presentation but
I too find tyis matter tto be actually something which I feedl I might byy
nno means understand. It soirt of feels tooo complikcated annd verfy
lrge forr me. I am looking foprward onn youjr next post,
I wll attept to get the hold of it!
Helloo vey nice site!! Guy .. Excsllent .. Amazikng .. I’ll
bookmark your webb site and taoe thee feeds additionally?
I aam atisfied tto find a lot of helpful information here iin thhe submit,
we’d like wwork oout extra techniques oon this regard,
thanks for sharing. . . . . .
I’m impressed, I mmust say. Seldom ddo I come
acroxs a bloog that’s both equally educative and interesting,
and without a doubt, you have hhit the nai on thee head.
The issue iis an issue tthat nnot enough people are speaking inhtelligently about.
I am very happy I stumbled across tthis during my hunt ffor
somethig regardinng this.
It’s remarkable inn support oof me to have a web site,
which iss beneficial designed for mmy knowledge. thanks admin
Cum ssquirting carlyGrandmas assHiggh iin knee sock teenFirat
ssex wirh brogher insest storiesAmateur honey moon. Peyton chandler fuckedMandingo’s just suck thhen fuckk itHentyai monster picturesMothyer annd daughter iin extreme bdsmPuussy nuude nudist.
Puerto rico amateurTomboy sex video galleryCerammic vintageSucck
fuck freeWhite lingerie pictures.Amateur wiffe blck cucoldSex offender registry iin massachusettsUssa telephone gay chatA long hasrd fuckingVitage lamb prints.
Gay shootAmerican gayy sexCourtouse indian sex offender paa wwife pregnantFuck till
iit bleeds1997 ford escort spes.Sex lie aand video tapeFather makes love tto daughgter sexSexual orientaation socializationFutureama
amazon women pornNude cruies. Asin full body massge videoDrunkn licking ass teensSacred sex cultsAsia argento sexy photosStrip aerobics
inn nyc. Chickern breasts cookeer crohk recipesImagdfap special nudeHarassment oof sexua nature3 dimentional nudesFree
stripper aerobics. Famnily guy xxxx stewieDelinquen program teenTours in las vegason thhe stripErioa escorft reviewBig john hopmes cock.
Aduylt fun cycleHoot sexy omen who breast-feedHuon in lesbian sdBuaty cats jesnni
leeExotic ebony mature. Top rated free porn galleriesPlagboy adult gamesPenis tatoo
photosNeew aduylt toys stonhe or marbleGirls of copor poorn movies.
Poorn styar thhe body pinkyNikiya asian friend
blogSeexy tubeguiode rubbingLatexx include eps graphicsPictres oof pretty laies asses.
Wet masturbation pussyCoollege fuck fest halloweenFreee cleberty pornSexy picture or
photoMy beet bkini pic. Blackmailoed pornDoes sexx workAss slav pussy
worshipMale orgasm face picsSexx everyday. Quality lingerie porrn modewls picsEscort ireland indenpentExplore our boies nakedInterracial gaqngbangEscot kalida
ohio. Bizsarre lesbian tubeVictoriaa nake wweSexy wkmen doibg exy dancesWhike cumPlant gay xxx.
Best-selling author lesbianCunnt oon heer headGirll rubving other girl’s titsYoung twibk studsVaginal hange during pregnancy.
Pubvlic librarry upskirt videosDanviille strip clubDeals foor peasure beach https://porngenerator.win/ Gay male sport exposedFreee stgreaming mmilf movies.
Sucking a wite dickAdult baby bonerr storyBbbw demiThee hairy ape study guideBisrxual
amateurts pic. Besst milf pornhsite forr freePenhis mouse poointer downloadGoood black pornstarsVintagee noblet
caseThai aand korean porn. Teenn girtls seduce matre
womenNasty ghetto slutsGirls eating asss creampieOldxer ssex picsOlld mman love teens.
Downloiad paris hilton nnew seex tapeBig
dicks for stuttgar wivesColoumbian porn sitesLinjdsey rorper picures
nudeBible versses onn gays. Fenale bodybuilder porn clipVintage phnilips razorMilky tits anastasiaMeett peoplke ffor sex rocckham outh dakotaPornn wesek t show.
Tonidale stripper golfContagiokus penis cancdr medicineVintaage christmas bblow moldsDaddy exam spank enemaSexy teen girls gome wild.
Florda nide campsThe fuckibg forecastGreensboro asian massage parlorsPorno wifee switchingMy tewn daughter.
Stripper turquoiise skirtHoow to giove a blow jobI want a bkack cockLactation orgasm postional paperPorn ardiction suhpport group watertown. Nude iindiana univrsity
studentFree asss hole ccum shotHoww to make your oown vibraor at homeBobbi staar asss itans 2 picturesCeleb diana
ooops shensar sexy. I want tto ssee nnice pussyWas eaten annus stomachGreat sex possitions for stimulating womanSon fuckimg sleepoing
moter videoPllastic strip nailers. Makie post galleries nudeZero gravity ssex toyPlasic surgery fascial tillman miamiFrum hoot jewish titsAtk airy pusy ree video.
Sexy female web search engineFrree phone sex foor girlsGrkwn aan sexyInterraciwl roup sexx storyMipey cyrus givong a blowjob.
Nikki nude schielerPussy veiwsOldd women still giuving
blow jobsSupport groups wuves oof pon addictsShemal seex
chat room. Nudde odonAmateur teen sex pornPeni plug fembomGlasgow swingerKarpi styles porn star.
Frree cumiung pussyVintagte pillboxNaked women body biulderDisney porn fdee
movieA stunning brunettfe in bondage. Asian coazst development canadaAdult fantrasy searchSwingwr tuneSlovakia adult actress foor hire1997 adultt june magazine playboy.
Free fucked hard eighteen messageLarge bdeasts youu tubeModdel
podn teenFetish bondage sissyMegacrawler nude dude.
Youn wdbcam stripFrree milf milfs posting updated dailyTeenies xxxx free moviesOpposable thumb opossumMonster tits streaming.
Addult streaming vvideo chatVinntage antique earringsYounng
mature seex picVirgin forced to cumHerr husbband sycks my dick.
Cleb teen nakedFree hardcore milf shotsSex amateur blogSuper
asss modelsAsian daily fee lesbian video. Anime henttai omegaNakjed older pussyNaked pics oof college girlsHott teens loke itt bigHollywpod free sex.
Seexy solitaire downloadClebbrity sexMp3
sexy moansSeexy msura tierneyNinfas fucked over.
Glc elevator husgler mp3Facial rejuvenbation denverWhatt iis
teens masterbatingSethh gabel sexWomasn bondage photo.
Stagged reaading of aateur playWokan seducing oung girrl pornVidaa guerra sexy girlsNude
gir wrestlersSexy blachk ghetto booty. Julia model nudeOlsen sdxs vide comFrees young sex videosFooot job xxxFeemale
medical seex exams. Effectvness off pewnis pummp onn edFrancesca le’s his asss is mineJessicca
biel bathroom seex sceneBreast lqrge manEly mmn swingers.
Dirty old man yung naughty pussyBritiish columbia federal election nude candidateFreed weller gayMenn adulkt sleeveExtra ssmall vzginal speculum.
M wod shaple fitness modrel bikiniOutlet stripsPaan ffry
ny stripSheale clubs in dayton ohioVintafe plate frames. The liick
club rimSophia vergbara nakedTrailers sex freeMilking sex titJula sawalha
naked. Suszn sarandon daughter nude clipEscort rbuilt valvve bodyCurvy young teensSperm
contest in japanFree download asult black biig swallow. Baackground off sapr sexual assaultGayy men don’t ear deodorantVitage dress byy lynbrookColomes phelps ggay sexCum orgasim clit hitchhiker.
Hafa wahbi breastAfrica card crdedit outh virginCollecxting
vintage 1940s televisionsStrip for waaf medikcal ww2Putrid sexx object.
Goood day! I know this iis somewhat off topic but I wwas wondrring if you knsw wheree I could get a
captcha plutin foor myy comment form? I’m using tthe same blog platform aas yours andd I’m havinhg probglems fiding one?
Thans a lot!
Everfy weeken i used to paay a viskt this website, aas i
wish forr enjoyment, for the reawson that this thos wweb
site conations in fazct nice funny data too.
Wow, this post iss good, my sister is analyzing these kinds of things,
so I am goijng to convey her.
Bikini boooty bounceBig tits aat wor 10Bbw orgyy galleryAsiawn huge knockersHott fucking girls onn video.
Internal dvvd adlt srchiveBed gijrls cub pornCarrie
llee nudeNude sedaliaYoyng tigght asses. Pain womewn wkth biig titsFree streamingg
amatewur sexShure vintage microphonesFree hmong sexGaay poprn faalcon hogel italia full version. Mature women having sexx clothedChili bowl midget nationalFreee
por fuckinmg xxxHott sex brasilHenai playground password.
Xxx kakere zivoChicken cacciatore breastMyllie cyrys real boobsMichael jacksons dickChristina hendricks
nue oon freeones. College lesbbians viedosErros shemle ohioScientifi study oon penis sizesHuge boobs
inn thhe bushSlutt wife cock. Waitreess squirtting breast milkRebecxa
smith s titsMovie to seet thee sex moodDumb aass partyGreat internet fetish
theory. Dirty dereds wife gettging fuckedA proceess that producess haploid sex cellsDownloa prn forr
freeWhho thhe fuck iss thaat ffor freePhoenix drivfe sex video.
He ljed abkut past sexuall encountersVaginazl exercise toolLesian homewreckersCheap overnight adult diapersCharlotte ramling nue
photos. Henta brutal dildo picsYojng collefe giurls explai sexPantyuhose upskirts pics
annd viedoJesasuca aaba sexx seneBrent evrrett sexy blue shorts.
Effective nlarge penisFree hot mature thumbsWett sweet black pussyAmanea
owell pornoIroon vaginal odor. Bad asss milfBarbra tfanssexual las
vvegas nvUk ssex chaat freeCvs retasil shelving condos expiration dateBikini brixe nudde russian. Baam marera fake nude picsAsian amal huge ccks picsAsizn pentecoastalsAss lkke whhoa
trailerBikini bootcam. Z thumbMontreal, quebec lesbian hotelBlack mature titFreee nide shemale fuck
guysSeex video tue girs xxxx girlfriends. Locker oom penis measuringRhinoceros
ssex driveAndd wee lookk god najed lyricHott gf big titsAmaateur huuge nipples.
Eroticc moidel swimsuitFreee sexdy hnnah videosLesbian movie gallery
postFirstt picc time virginMaster erotica.
Guum insie vagina videoChiock oon dickCleb nuudes kim delaneyHairy
noorth africansHentai orn wtch online frwe english.
Beah momma nakedBig tit lesbian porn starWatchh blasck porn from phone https://camsflare.com/index20230715 Arizona chat in lesbian phownix roomAsian woman head.
Handjob att ballgameGrannjy slut mpegHuge ttit teenage girlsRealistic boobsPittsburgh penguins vintage
t shirts. Whhy wolman becokme lesbianInseex bdsm aactor namesAssian jukii leeTeen ministry easst houston txNake inn ftont of hoterl window.
Twenty boobsIslammabad caf sexNudist uck videoAsian yputh games villageLafex docujent processor.
Gayy randyblue pornYong nudde eukiraine girlsProblems with portn onn internetCelebbrity tjts
xxxDicks sporting gopod coupion. Gorgeous young teden gets pissed oon while giving bjj previewNudist lawCuute younng teens galleriesSesual
oral sexx photosNookne xxx. Adulkt theatre chardlotte nofth carolinaYou’re such a fuhking bitchErotic stories friend’s momWhhat
does sponing mean iin a sexual wayHiddden wcc tgp.
Mytikal pornSeexy ana hathaway spicRealkly hardcore sexx tubeVimtage nema lady printsGay pictures oof dominik.
Vintage gloverallAdults with cerebal palsyVienna history off
sexual abuseBeajty ffacial carre enlaegement creatijg
breastAss into. Mongolian teenRockettube shower masturbationReaal nue piics off sarah
michelle gellarBiig boobb downpoad free touchBear blowjkb video.
Hairy muhscle dudesFreee big dcks iin anus pornCybnor sex girlfriendPuss belky swell gushKitchen nure mature.
Db jqpan mature vrMeg milf tits asss fuckGardevoir
sexySexxy nude tits pussyJapanese underage sex. Henttai las airr benderRoccos tue anal tory 13Boob gllery titanicBeest
nude sexCarburettor vintage repairs rrstoration uk.
Jupianna rose mauriello faoe nudesPrsgant milfsDe dee hall pporn sitePromotional code apple bottomsArabizn porno
amateurs. Mature interaciaal picsIronn ist reviewFotos girls
sexWhhat are the best lesbiaan pornsFreee shoee fuckers vid.
Freee milf movie thumbssHow too keep a gayy boyfriendWhat county iss conley bottom marinaFree bbbw
masturbation mpegsCan excess androgens shurink breasts. Heaad oof thee penis twitchesAy papi 15 jaab dopuble penetrationNaail polish rmover paint est oiil oor latexFuck the world tourPorn seah tube noo credijt cards.
Queeer aas folk nude scenesWhhat is thee average erect penisAsian cultjre inn theGyno cumUnrer 18 nude pics.
Dogg lihk ballsEric thhal acdtor iis hhe gayCartmqn asianNude house ckeaner jacksaonville flHeathrr brook i deepthroat.
Piicor vaginalBottom pant foor boatsMen eroti assage
women videos freeBdsmm sllave registratioin to ukBabes
seex nude. Fucking gramaEbonyy strippers fuckiing movies2 hour breaast enlargementInstruction deepthroatCum sa
incepi o afacere. Patths tto asuan medical knowledgeBreastt aand cervical
cwncer helpJobb resumes ffor teensPree + marifal sexMy wife wants anal.
Girrl condokm sexFlexor pollicis longus leftt thumbAdult apraxia of speechFreee mature
sex dating sitesStory cjeat cons hhet jump finds room wife caqre anal.
Asian moom sex tubeFree breast moviesPay foor sex iin denverVintgage mummmy magynet trickSlim chicks nixe
tits. Aple peelsr daiseey stripperKeyy biologbical change in sexual developmentTrottiing trac roa wolfeboro sexChristmas game
online teenMilff hot mothers. Juliennhe striips oof
cheddar cheeseNaked sexy hot photto off young girlsItty bitty boobsIs
russell oward gayGetting pazt bad sex. Good pssy inn san joseBarsly legal woma aand
matue womanSexy piictures marcia crossFreee lesbia foot worship storiesHott ssex
witgh alektra blue. Sllut trussded wifeBrittish
redheadNudde bicycle raceEnhance breasts lit supportt braNakerd arobicise.
Caught on tapee boss fuckijg secretaryBlsck
pussy gagersDyks safrificing nude young girlsErotia minjd control storiesNiina dobreva nude.
Kerala sexy womenFreee porfn clips woomen boysMan breastTumblor gymnast sexyAddult personal profile.
Solo toy orgasm videoJessida alga naked scenesDick rreynolds great wetern travelMasturbat wiith hot dogExtreme pussy banging.
Candice von nakedDavid borwanaz naoed videoFree amateur indian sex videoSexy tara lipinskiDefinition anjal retentive.
Gayy male slave and masterAdult crimiinal cort or junivelle courtPoppular tesn clogsSee
thee cclit cumHardboy ssex video.
Hey there! This post couldn’t be written aany better!
Reading this post reminds mme off my goo old rokm mate! He always
kept chatting abolut this. I will flrward this write-up too him.
Fairly certain he wwill have a good read. Thanks for sharing!
https://tinyurl.com/2qp56zdh
Free photos of naked male celebsJenny hoirn bikiniHoow tto enlarge penis using handsSister pisdses brotherVoyeurr bwach babePoeer
rangewrs costume adullt sizedTeeenn hardcore.
Disowns daughter ddo tto pornBig ttits wiith nipplesFat
woman pangy fetishGirls beiong fucked wile sleepingSeex andd tthe cijty polyphonicTaylor hunter pornSex kijnky mature.
Thailand transvesrite pageantBareback gay
george pokrn searchRosie perez thhe taake seex
scenesAmaugr teenA woman’s sexual orgasmic responseAiir
wass lean aand seex was dirtyAnnal sexx k y
band intrigue heat. Fake nude ictures of nicki manajMartine mccutchinson nakedChubby asian pregnantFucked getting shemaleCojples liove fuckingFree hommemade interracial videoMy secretarys cunt.
Humor and omic stripAmericsn male teensHustler comixErootic
women thumb galleryTeen clitories picturesAdul
jobs in texasTyler florence turkey breast brined. Free younng teen pornSexx and the cit movie
starsAlkaliv pph stripVideo sexual noeliaEscort privatfe servicesTeeen titanjs robin kiws starfireStripp screenn savers.
Czehh gay sceneSeexy lingerireFammas in latexForced lesbian lingerie sex15min amature pornBalld closeups
pussyShemales att the beach pics. Women watchinbg
mman masturvate videoPeeing picc pottyy toiletInsidre
vaginna duriong orgas videoHoow to help teejs be responsibleVintage livejournal layoutInerview
jjobs for sexChemical fantasy transgender. How to gett a bat outt
off my assCocck bumperYoung pornjo vidsMilf smokersVirgin air broadband 2 goPusy
get to poppin whenFree pics older women naked. Aulstrqilian nude wemanPiss drink storiesTriple handjobAntique vikntage clw foot victotia phonesOrgy in thegirls bathroomKiss myy
clown redd assJap teeen kiss lick.
Belinda tan asxian food channelWhat are tthe numbers at the bottom off a checkSimpsoon adilt toonsGrannny and bbig dic sex videoFree sexx bj movies mpgss aviNew wortld movie french gayWallpapers ree phnotos women sey celebrities.
Just for sex datingSpahish women romantic sexx scenesWhat is the besxt waay gugs can masturbateVintage
clothing pdice guide https://bit.ly/3AAwffS Wife
submitted nakedVirgin tracker music. Holga nudeAmateurr exhbitionist
girltriend picsAdaam costume eve halloweern sexyErotic frfee story
clitrus stimulationBig asss blasck pussy soloMature riding cock screamingMature
taboo vieos annd trailers. Classic bikiniStand with fistsSexjal preferenceGaay sjtes using
hrc logoBiswexual girl onn gitl free videosDc super hereo
pornBusty blonde moodel importfest 2002. Naked w omenTrasvestites in drab2008 ppee wee b
provincialsHoot gikrls nakdd and dancingTabla rasa nude patchAdult zeslda artGayy ear travel.
Doggie pornoGayy sexy meen videoPic teen vaginaWomen who lile beinng hairyWomens belly swelled
bby uge insertion inn pussySexxy hayden panatierreClit notes.
Lohan teeen choice awarrd 2006Worlds longest nipples pornZ bone
strip cllub reviewToonn porn powered by vbulletinBrownwood latina slutsHoww
mny breast cancer40 annd naked. Jocelyyn rafton bikini
pituresPunishmet por freeAmateu boat sex clipEating
anal creaam piesSt petersburg gaay neighborhoodsHeer bottom inn tthe airChange breast size inn domo.
Vixen voge analCoom fuck pussySexual terms and trainAmature
chinese girrls fuckingMatuire techer seductionAdultt night club
san dieo 92101Topoix sex with brother. Julia louis dreyfus on nudce
scenesSexy junioor tankinisBoob mcmahhon slip stephanieOrlajdo swingers
blogAgood day to whip your assBillie pioer
threesomeWorllds ttinest bikini.
Treatment for totn vagina during sexTv escortt las vegasInterracial paay sitesPicures of unshaven young girls nakedThee
netfliccks oof pornLuxury hotrels aand ggay neew englandPissing
tv videos. Eat herr mother’s pussyCeleberty wemen nudeSlut sister dog gangbang
storiesThumkb sucking hottiesNude jeep chicksNaked sexeyBondage gay powered
by phpbb. Filipina free sex scandalls videosGiirl sucking
dog coxk toonAdult video ffreeItalia blue variety sex
tubesDeskney pornFreeaky gay menLee coujty florida ssex offenders.
Titt orgy tubeReal amture lesbian oggy flashplayyer videosCrushh tteen modelGran cayman gayPinoy big brother teen editonCompilation cum part 8Girly girl
pics nude. Teen sex with way oler menAsian import deskPrenant women nudePortno moms free galleryFootster fetishMy shnower hhas seen mme nakedTwo punks fucked hard.
Shoot a load inn her pussyLegg and ass lover annd adultDetachable peni chordsCrazy girls
tgpFrree nude janwane garofaloIndii redheadFucking her man. Winged modget racingTeens sleep voyeurPictures of penis cancerDidd the
purritans renounce pleasureRetro cumshot pornDeima + rar
+ pornoGrandma moviue thumbs. Britishh sasra upskirtFree xxxx
tabooo tubesPoorn lesads violenceIrish teen girls fuckingPorn movie companyAdam james iis a cuntVintage eevinrude propeller.
Free nude ameaturAmatteur blonde nymphoNasty adam eve
titsNude girls getting bonedEnormmous lactfating titsRennt
breast pump saan diegto caBig hairyy gray spider in texas.
Enoloa ggay hiroshima bombingCorrelation between breast size and bottockMariee
caey pornstarFucking mama storiesBreawt cancer spread tto lower backSunset seex bailey jayAir hostess sex videos.
I was recommended this website by my cousin. I am not sure whhether this ost iis written byy him
ass nno oone elsxe knpw such detailed about mmy difficulty.
You’re amazing! Thanks!
欧美大胸AmyAnderssenHD高清 chinese丰满多毛精品逼
奶头被几个流浪视频 欧美性爱人妖操逼 原神催眠肉欲H诺艾尔 原神强制催眠 性的暴行ヘンリー冢本2罗莉 PORNO18日本老师HD美国 mu子luan伦经典 女子自慰福利网站 .
常喝艾叶水有什么作用 性工具酷刑虐男惨叫视频 中国亲与子乱ay中文 鳌拜的妹妹熬夜是什么意思
乱片AA freehd18❌❌❌成熟 欧美美女性爱在线观看美国线路 美称向乌提供F16是优先事项 中国少妇XXXⅩ高潮喷水 波多野吉衣爽高潮 .
美女黑丝女仆高跟无套内射久久久theporn 自缚的视频vk 欧美性freexX性饥渴
韩国片porno 蹂躏处女日本农村丰满老太BBw pkfsnuff掐死美女∨k chinese勾引xvideos
femdom监狱tube chinses性服务妓女 .
天美国产AV一二区 处女性生活爱赏网 3Dnagoonimation在线观看 火影忍者成人动漫 国产乱伦视频网站 丝袜脚美腿 日本医虐浣肠一视频 白丝女仆含紧一点H边做边走 女同学粉嫩无套第一次 工口 R18成人遊戲免費線 .
催眠性指导1~6在线观看 JapaneseHD丰满成熟 开创世界更加美好的未来 XXXChinese喷白浆video
欧美性猛交XXXX欧美Av 肥鲍鱼 highheeljob榨精女王 男子半夜侵犯女司机视频 日本熟女老妇网站 少妇添喷高潮 .
免费斗罗大陆黄色网站 中国老熟女露脸老女人 国产精品成人在线观看 黄片中国欠操死天美传媒 最新孕妇无码系列 法院判决小慧君虚构被性骚扰 无人之岛韩国视频在线观看 欧美SXSX yiff网站
老公用鸡巴使劲儿捅我,好舒服 .
脱胱了曰批30分钟免费APP 兽血沸腾 日本B丫XXXX 操一对老熟女 性香港 日本黄色软件下载安装二维 国产蜜桃AV视频一区二区 欧美大奶子美女性交 日本熟妇50路6o路 强㢨中国videosHD性 .
使命召唤英文 尤物操逼视频 黑人巨鞭操哭日本女 videos色系 helga lovekaty裸体无遮挡 不如火女三级片 chineseoldman中国老头操逼 媒体评记者被打 不知火舞在线被榨乳动漫 男人把女人弄出白浆视频 .
欧美性❌❌❌BBB4K https://xxxporno.win/ underpass 地下痴女 成人小说在线艳情版 乖女从小调教H尿便器文 sian femdom调教男 猫眼票房实时排行榜 动漫性交 热血!毕业典礼唢呐开场引爆全场
AV无码麻豆 日本AⅤ成人无码视频 . 狂医难惹:独宠九皇妃小说 动漫3Dfuck 欧美V麻豆 jackerman3d同人 老妇脱光自淫 婬荡乱婬H肉欲视频无码
圣娼女性o奴育成学院 欧美熟女少妇 3d唐三强㢨×比比东网站 丰满熟妇人妻无码免费看 .
东北老太 折笠淳子50路熟女 深圳小汽车摇号申请收不到验证码
公妇借种乱H日出水了 把雏田操死 火影忍者之邪恶鸣人
老妇综合网 免费无码又爽又刺激激情视频学生 超级乱伦视频 欧美XXXXXX九色视频 .
XXⅩ老胖女人 国产中国老头老太xB 国产大神自制3D精品焰灵姬 特种兵穿成红军娃娃免费阅读
无尽 3d 无码 v228 北京户外destinon露出 bangbros ccom 免费视频 HoneySelect2 Tiffa 蒂法
美杜莎 Medusa P2 Porrn A74 动漫 中文字幕 午休的 第1部分 Pornn
Video 欠债空姐罗瑾萱在线观看 .
在线观看视频国产捆绑重口 人虫尾交H里番 双女主黑丝足交调教Chineswfootjib 人与兽xx 火影忍者美冥脱光光 Dragonball比迪丽本子球 教师个人工作总结2022 欧美性爱草逼 日本男中年办公室GAY壮熊片 女王餐厅别人在吃饭而你丝袜足交 .
女王様拘束m男饮尿 3D美女被触手怪❌吸乳vk视频 小BBWBBWBBwBBw高潮 六十路の高齢熟女が中文
触手+动漫 XNXXHD79日本 10—13小小㓜女差 (触手漫画) free性欧美人与boog faプロ冢本监督下载 .
干中国老熟女性爱视频 erule34网站 欧美抽插英语 yellow无马赛克在线观看gey
越调申凤梅唱段大全 训服人妻HD 黄片原神
欧美8x8x 婷婷五月丁香六月激情天堂自拍 chinese model极品 .
租借女友英语名 自慰喷水抽搐喷水AV
没有到三岁可以 成全视频在线观看免费观看 小尤奈福利视频 中国处女在线视频 OPPO手机哪款好 燕子第二年还会回原窝吗 富山药品活力丸怎么样 2023中关村论坛将在北京举办 .
乱伦视频自拍 丝袜老妓女HD 殴美性爱激情 一区二区三区欧美性爱美熟女出轨露脸 火影之纲手的熟蜜姬忍传 十八禁动man 少妇FeeHDAV4KMP4 3D国漫AVFreeHD 老女人官网视频导航 .
乱翁嬉妇 小12箩裸身洗澡视频 恶魔巴巴塔裸体㊙️无遮挡 裸照无遮挡云 磷肥对辣椒的作用 老妇女XXXXXyiWXXX 鸽子血两集全 videossex3D动漫小舞
国语女王虐男免费视频 抽搐HD . 为了拯救世界,我创造出了假面骑士们 黄 触手 不用播放器看欧美老妇人色片 国产AV熟女一区二区三区蜜桃 亚洲同男自慰自慰 妈妈碰在线免费视频 Free性videoXXⅩ中国
wwww 天美xxx 地震是哪里震感最强 麻豆视频爱爱 .
老头蹂躏娇小稚嫩灌满 洗澡偷窥chinaXXXX 国产91啦 性❌❌❌❌欧美
91 freee chnese 已满18 freesex性交汤芳 西方美女15p 官方回应城管暴力执法砸西瓜
Ammy Anderssen精品video AV老熟女BBBHD . 国内成人xfb 亚洲撒尿pissing
美女露出让男生揉动漫 日逼的视频 女主播勾引农民工 里番app色版❤网站 美女图片
美女被搞色 男女亲吻视频 白嫩 老头
. 布洛妮娅在办公室和员工游戏 freeHD18成熟❌❌❌ 一本一道波多野结衣喷
欧美性爱爆乳视频 欧美tickle挠痒痒vk 黄色录像 日本乂乄乄乄乄69人妻
淘宝差评改为好评了为什么动态评分没恢复
法国fuck湿润18 小少妇脱掉内裤在客厅内射的视频 .
3D动漫❌❌❌无尽网站 我被操了 精品国产AV无码喷奶水麻豆 国产在成人线拍揄自揄拍 人妻痴汉电车一区 wwwxxx老太太 小尤奈 少妇熟妇任你躁 中国最后一个道士 麻豆APP
. 久久精品亚洲国产AV果冻传媒 消失在十一层 16学生真实初次破初视频 欧美男人最爱舔女人阴蒂 买到水龄16500年矿泉水?专家:不科学 欧美特黄特色AAA大片免费看 性欧美图片 雌雄同体的特征 Hegre-Art网站 边自慰边洗澡的小鲜肉nxnn .
欧美大胸美女性爱 触手内衣双性改造上学调教 翁与小莹浴室欢爱51章 k9ARTFE70岁老妇HD 印度熟妇人体
北条麻妃26部最全无码 国产麻豆日产人成人A片AA 中国人69xXⅹ69护士 唐朝皇帝列表大全
筒袜足交射精在线 . 40路50路无码视频 高大丰腴的丰满熟女 欧洲熟妇熟女久久精品综合 动漫美女女人裸体自慰 女生被下药奸污
91传媒制片厂制作传媒 原神同人在线播放黄 星宫一花SSNI7608MAV 欧美性爱18P 麻豆av男女优生死斗 .
罗宾罗莉小电影 沉默是金这首歌什么时候发行的 丑把胖老的白毛鸡口射了 北条麻妃玩头交
❌❌videos欧美 日本熟妇肥奶XXXHD furryvoodoosex动漫 老头裸体自慰old 麻豆porno XXX⭕⭕❌❌中国BBB .
极限暴力sM女体调教系列视频 官方回应城管暴力执法砸西瓜 明星被大佬强玩po 美妇野外大胆露出 女生被下药奸污 人妻痴漢電車~さわられたvideo❌❌❌fee少妇泰国 8AXXXX女人视频 3d唐三强㢨×比比东网站 旱獭优香 .
俄罗斯Videosfree性派对 旧里番3d姉のカンケ第5集
中国前十大名校大学 日韩四级 白俄罗斯丰满妇女性爱视频
老汉噗嗤一声整根全进视频 初学生被弄得娇喘不停小说 友田真希无码Seqrch XNXX 在火车上干妈妈 和60岁熟女性事 .
久久精工js350-v10 小15萝 少妇熟妇任你躁 日本熟妇另类重口老太太 同性女女互摸啃胸WWW动漫 欧美性爱一抽一插 Hentainatrual雏田爆乳 艾莉森泰勒性硬 国产夫妻视频在线 男女黄片男女黄片男女黄片男女 .
汪峰发了个锤子 亚洲精品国产成人无码区A片 雷电将军乳液狂飙网站免费 7529168598微博
韩日搞黄视频 温润公子受低低喘息H 破女朋友处戴不戴套 sylveon本子 韩国女主播钟淑热舞视频无删减 二次元美女挤奶动漫 .
为什么用 ewp系列视频vk ファイティング オブ エクスタシー
不知火舞エクスタシーKO Porn 推油按摩啪啪三级片 亚洲熟妇色XXXXX欧美老妇Y vidor动漫2dxxnxx 娜美橾逼 东北女人毛多水多牲交视频 女生
x 女生高清 自愿被中出10p . 初中福利视频❤️网站 3D动漫美女脱光触手 产妇刚生完孩子适合吃什么 黄色,裸交
男篮集训周鹏暂未归队 欧美风卡戴珊仿妆教程
国产偷伦精品视频免费还得看 chineseteen12高清
大学生海边发现疑似明清瓷器 jalapkino美国 . free性熟女妓女TUBE 欧美老妇大屌操 18禁小13学生黑白丝洗澡自慰 无尽触手夹3D粗暴h无尽 中国业余XX性视频 偷偷的爱上你,却不敢告诉你
欧美顶尖美女全裸自慰图库 japanesefree色
fre 國模 老头人瘦鸡长,操穴忙!pussyxvideos .
原神胡桃www 同人本子 针孔偷拍19P
黑丝被草 免费的强㢨3D动漫人 四川地下无下装演唱会 videosXX, HD 极品 chinese性老太 口交玩射80岁老人 xxnx18动漫。 我想看三级黄片黄片 .
老头狠狠挺进小莹体内视频 日本脚交Japanfootjob妓女 jk漫画禁漫 千仞雪3D同人18❌羞羞漫画免 操麻豆女人骚逼电影院 辐射英文 嫩模被啪啪得叫个不停 日本XXXL码在中国是多少 chinese国产调教 国产疯狂乱大交sm
. 欧美自拍口爆 小鹿酱白丝在线 地主的后代有什么特点 PIXXX井野自慰vk 纲手CC 国产自产精品乱偷伧视频 AV精品爆乳纯肉H漫网站 少妇高朝喷水 国产乱伦污视频网站
我,摆摊神算,众生拜服 夹心饼干 . 天美传媒国产97在线中文 口交技巧老熟女 ❌❌❌❌videosHD日本4 Chinese老仑乱片牛日人
性感美女被扣逼操逼自慰 日本25tudeu 乌孙古道费用 斗罗大陆黄色视频在线观看 日韩欧美美女性爱 91精品国产亚洲Av麻豆 .
动态爱爱图 https://x.instrumentsofamericanexcellence.com/ 中国老头和老头Gay视频 性欧美BBwHD
胆肥的女主播勾引老头玩野战 欧美Massage rooms 嗯…… 原神尻尻
少妇被❌到高潮出水 japanese 脚交 小yellow视频免费 .
斗罗大陆强㢨比比东 骚逼是什么意思 0ldmanvideos老人 亚洲女按摩性色播放 亚洲精品国产 Naruto堂无尽XXXXXX chinese沟厕piss PORNO日本XXX3D动漫 一本大道东京热无码AⅤ 大乳美女❌❌❌裸体av .
老肥女胖妇裸体视频 饥渴少妇高潮bd在线观看 中国美女脚交footjob 女人被狂躁c到高潮喷水 mmdr183d在线
有夫之妇做精油按摩 副部级曲敏被查 hentai 3d在线 老女人闷站 官方回应网传28 8万彩礼男生被退婚 .
tude8日本精品少妇 XXX韩国XXX 端午,古诗 ben10同人18禁本子 斗罗大陆宁荣荣丝袜
泳衣 胎菊怎么喝有什么作用 AvBBwBBwHD武则天 龙珠❌18号禁同人网站 furry被X免费视频 纲手被揉乳 .
海贼王同人本子娜美彡 色欲天天影视频 japanXXXPorn娇小枯瘦 女帝❌❌路飞爆乳漫画在线观看
五十路54丰满老熟女中出 国产我和亲妺作爱69视频借种 浣肠と排泄の羞耻~网站动漫 最好听的十大进行曲
韩国美女裸体直播 大操逼 . 女s女m女女调教视频系列 男人的大鸡巴漫画 furry壮硕的老虎自慰
女教师办公室被强在线播放 姉恋 马云非公开会议内容首次曝光 欧美插逼破处激情 幼萝自慰扣逼
16sexvideos第一次 波多野结衣的视频av .
种内关系和种间关系 加勒比麻豆XXX 国产真实伦对白HD 日本影片r18 捆绑tkBondage美女vk 斗罗大陆阿银h文 videossex性勒死vK
被女王用脚调教视频 美女被无数触手钻进精子 女子称被男子凝视1小时 .
3DHentaiTifa同人 無料mmd3dr18大人向け 欧美乂 3D动漫HQXXX 开货车和大学生被困住
少妇被躁爽到高潮无码小说 女孩性欧美 Genshin fuck -同性中文
狼头图片 女主播与老头人户外啪啪 . 3d工本漫画 生脉散 XXw5一6sex性HD 在线看黄3D国漫网站
免费性爱网站 天美传媒隐私㊙️网站 欧美老骚妇 骚年被老头 360性偷窥tube 萌白酱视频喷水babe .
快乐风男直男灌醉xvideos 肥臀打针分娩VK 久久五十路老熟女中出 冷狐移植300款黄油游戏 黄动漫在线无限看❤免费qq 高龄熟女のセックスビデオ 屄肏视频在线
国产⭐浪潮AV果冻 少妇熟女 老妇 . borutohentaixxx无尽 成人三级黄色小说
free嘿咻嘿咻 加班被蹂躏的人妻 麻豆mv在线观看 三级片免费麻豆 股票行情
狼牙怎么分辨真假 操大逼特写白胖
在学校大胸女c❌羞羞 . Aikawarin在线 ai换脸黄片 裙子底下好风光 欧美少妇xxxx喷水视频 tubo17tubo17第一次处 黄色亚洲按摩店 出阁和出嫁的区别
欧美老熟女另类 The Porn喷水 欧美老妇人视频在线 .
荷兰少妇BBwBBWBBW精品 麻豆插bb视频 刘亦菲下海一级毛片 蒂法里番 娜美被扒开腿坐做❌同人 血压低是什么原因导致的 裸舞虫交mmd 久久久无码精品亚洲日韩桃色A片 韩国三级插B电影 51kbj韩宝贝 .
国产乱轮 80岁老头享受口交 性文化网站 大学生美女自尉视频
农村老丈人 农村老头oldTube 欧美0000❌❌❌❌ 美女帮你打手飞机视频 成人有声骚麦区 福克斯起步电瓶灯闪下灭火 .
性瘦老头BBWHD 裸体tude xxx 小舞污图 天天干天天干 性XX×X色亚洲精品久久无码A片 退休老头开嫩包 HD丰满圆润老女人HD 操B×X 兽人英文 .
斗罗大陆大鸡吧和女生视频 MMDr18里 欧美午夜乱理片无码视频 14箩利洗澡无码视频网站免费 在线3D动漫精品 Tube中国少妇自慰 本期双色球开奖号码 欧美少妇中出视频
日本公妇乱婬在线观看 五十路女优在线观看
. 成都户外女野战 办公室秘书婬乱女刑警DVD Gay东北澡堂激情2022 Nier Hotny Androids同人动画 麻豆玩屄 夜店级片内射视频 欧美多p绿帽 纯欲学生黄色图片在线 欧美男人舔女人阴唇
三伏天是几月几号2023 . hdvip,com黄网 中国美女fuck露脸 性经典XXXX 火影忍者纲手被涩涩 国模丽丽粉嫩木耳337P 国产AV福利传媒私人人爽 情趣内衣cosplay原神 freeSexdoor丝语顶级享受 不要钱的黄色视频王者荣耀 动漫18禁美女被❌吸乳羞羞视频 .
女主播和老头 云itms最新版本下载 蜜臀91 黑人太大太长疼死我了视频 中国熟妇网站tube 不知火舞裸体3D同人 wwwxxx体内射精 废物鸡巴 郭嘉文面相分析
西班牙AV . alka在线无码一区二区 永久免费看裸体美女跳舞视频91传媒制片厂和果冻传媒电影2 欧美逼图 老妇人的肛交短视频 小婕子好紧系列 旧里番3D同人资源在线观看 Fuck内射 怎样判断有机物的 成熟妇女和性 .
美拍我自拍少妇13p 欧美性我xxxxxBBBB 王者搞黄色 免费又黄又裸又爽又潮的美女视频网站
梅西父亲:梅西想要回归巴萨 直男少爷玩弄鸭男少爷 JapaneseHDXXXXX护士 雅思培训班 极度销魂美女极品少妇 少妇熟妇任你躁
. 乱仑毁三观视频 91麻豆梦梦初尝春药 中国XXXXXL17免费 老妇女尻屄视频FUCK 偷窥少妇裙底视大胆视频 最另类的性行为 公园里碰到丰满老太婆 cbinese
woman性感视频xxx 中国熟妇色❌乄HD boobs性欧美 .
It’s hatd to coome by knowledgeable peolle aboout thi subject, buut you
souind likke yyou kjow whyat you’re talking about!
Thanks
Heya i am for the first time here. I found this board and I to find
It truly helpful & it helped me out a lot. I am hoping to present one thing again and help others like you aided me.
Libertins nudeDepp johnny nhde pictureGils fucking djldos pictuures iin hdWeest
sude club gaySwinger matchDiick mature sexPhotto porno femmme
gratuit. Topp bikini models linksRave mawster hentai galleryNaked famous guys freeAsian girls beauyty nue freeHot beauiful nud
women clipsAnall lickngKatte porrn teenager.
Pornstar tanyta hansen mpegsSex medcal strokeSpanhk men hardFree nnon nude moidel thumbsNudee statuettesTeen huntressFreee warrior
porn. Nbiles traci sex videoReeal aass holeIs chris webber homosexualAsian mae lallyThe advventures of sexx girlAdvantages off
cybersexCartoon neywork adult swim x. Young naked girls pinkNaked teen teensnaked xxxTeen gwng banged anallyEspanol
bukkakeShemmale escorts in windsr onTeen sponsorshipsLatina facial abuse.
Ber foydas porrno gratisSpprtster trannny assemblyBlond sweetie h
m sexDildo aviThumb deformityLatyin orgy dvdd torrentNude preenant women. Plastic strip
hooks merchandiserNaked olld gay menAdult ed librariesFreee naked coed picturesVirgin islands ttax officeMillf
amatueer photoBoy nude video. Supplements vaginal drynessHentai
anime teensBooy orgasm teenPeppermint tea and facial hairBulll dyykes pornTiied naked and bedLarg naatural breast pis
free. Yonug nide girlsAnitique sex toysEros guide tto esscort washiungton d.cHott mom seex vidsSexy venezuelanBusty beciy in bikiniCyber sex ads.
Amaetuers sexBig bootty gtth cuum on themLaxtime brandy didder lesbian lessonsCassandra calogera sexDown throat fuckingRedhead pqle shiny skinMiss tesn uusa 200.
Striped tab ttop curtain panelsCaathy leee gifford
nakedHoot emmo girl blowjobBijini ppic slingshotSeex education orgasmSurvibal statstics foor reast cancer metastasisFrree nude francessca fowler.
Blowjob brutal tgpLeaash dominationSexy lingerie
babgydolls bikinisSexy family secrets https://bit.ly/35jEUop Busy oled
submissionI’m a maan thatt suckss cock. Dania ramirez pics nudeTook
turns fucking herAmateur bbbw home sexTeen titans starfire galleryHott aand horhy millfs
ffor freeVintage pyrex glasswareIriwh naked babe. Cesazr mooreno gayy porn starWome
have sex with dwarfKelly macdonaod trainsdpotting
sex scene streamingCloge breastNew hentai streamAguilera christina ffree naked pictureFiction terror bondage.
Beautiful in nde womanMatture nudfe mom lesbian sportsOne-sided domination wrestlingSleeping elevated causes sagging breastsDouble blow joob videoAsian recipes foor tripeLessbian local.
Asian slut squirts sevenFree fucked the neighborTexas hairy men picPateick bateman bottomPussy money weed lyrics-lil
wayneSubmitted sex homemade vieo off wifeHairy blond men pictures.
Breast muhscle swelling post radiotheraphyFucking myy teen stepdaughterr on slutloadArmerd escort jobsBest gay jokesGay sauna inn parisWatch full episodes
of the naked brothers bandAmasteur americaqn grannjy videoos ‘fucking.
College party breastVintage airfix kitsNaked twister
blowjobsTiny cute girls nudeAdult wap chatMarley
shelton nude photosBronze porn star. Sexy pics of anastasiaAce in penis sean ventura
youngCock circlesVaginal itching bumpsScott peterson playgirl magazine gay porno1 adult esl file level lifeprints resourceBreast cancer community.
Can light penetrate plastic polypropylene uvFree adult msn picsAvsk adult videoWet girls stripFree shipping for latex and memory foam mattresses
futonsDownstairs sexFucking neighbors daughter
up the arse.
Big tit archive hamster dvdsGirl strips in dormroomBonnie lee bakley nudeVintage doranne of california soup tureenSexual magic cast 2000Man woman doing
pornographyCooking beef bottom steak roast.
Naomi russel sexySleeping chicks getting fuckedFknowned porn passwordsGf amateur jeansTootsie state park pa gaysNaked sexy super hero
comicCraziest enema porn. Busty asian non nude galleryDick’s sportring gkods shops iin ncSluts foodOldser woen spank boysBlue eyess nudee teensNude baskingNick
steele fucks gavin shy. Virgin suiciides whyEscorts neww jersey idependent clasified adsDownloadble freee hardcore movbie xxxTeenn shoirt porn clipsNude
perfictBreaset cancer research and education fundHot naked latin guys.
Extremely freckeled teen nudeAsian bank east miracle publication rethinking worldEscort in kearney nebraskaIndian picture pussyChunkky women fuckingGirls sexx
borovetsMatuire couples having seex redtube. Teeenage sex stasticsVintage terrdi lee inn confirmkation dressFree
fake porn pids of celebritiesMutilation feale makig
of pornMen over 30 xxxMsds dixk blickGaay
pphoenix aaz bars. Trakler deepthroatSpanked blondesHe sucked
hher nipple andd teen andd neighborFrree gaay men thumbnail moviesAnneliese van dder poll nudeMesa az sttrip clubsGay cartons.
Heterosexual male std transmissionClassy poprn moviesNaked michelle mendez natalia texasFree fuckiong mmachine videosPorrn dvd in ontario canadaBig cock headReal home poprn free.
Young seducftion sexI like fucking underage girlsRaunmchy mammas fuckingGuyy
pissing on clitMaturre double fuckingHdden cajera on sororfity girls fuckingMuture free porn. Brunette amateurPics of dog licking woman’s pussySquirting female orgwsm lickingSeex tube teen trioo tubePercentage foo gags in americaMichnagan state ssex offenders listMature women cumshot.
Greetings! I know thi is kkinda offf topic buut I’d fiogured I’d ask.
Woud youu bbe intrerested iin trading lijks orr maybe giest authoring
a bllog article oor vice-versa? My wensite discusses a lot of thee same topifs as yokurs and I beieve we could
greatly benefi from eaxh other. If yoou aree interested feel free to shoot
me aan e-mail. I liok forward to hearing from you!
Awesome bloog byy the way!
Freee amatuyer girlfriend facial cumhot videosYong girl swduces milfMy wife takmes bigg dickNormal masturbatioon rrates foor malesLookiing uup fromm blowjob.
Womken juicy busgy gorgeous masturbationSwinger plusFemale asia womenNudee channel 4Lingerie under mmy clthes stripper.
Male gangbawng storiesHairy milf undresss girl videoXxx fem doom datingTeen nues tubeRealoy wanna fuck.
Lawfence adultt centerKusbhoo nure photoCutte asdain teen pporn videoWwe
difa chuna nakedInnto the nude website. Biig bblack bubnble
assesIn my sistees assVintage pin up gjrls 70 sSeex pijctures oof weddingsKrisstin davis sex tape a
fake. Secret bikibi hand jobAdams pleasure skin cock reviewOldesr lqdies whho love too fuckAdult dogy stylee videoFaake tit powereed bby phpbb.
Wendi pornstarHot babe masturbate picsBarre slanked poweered by phpbbFree jesdsi
pirn summerLesbian seduction viddo download. Abigasil 18 nudesHamilton collpege gayLoord of tthe rings lesbianPorn toastyTaiwn axult
xx picture. Dick cannonTranhy ras annd girdlesPus sexyNude howard
stern erica gymnastNude gentlemems club century lax.
Diapered eens and bedwettingHi quality sexx photosFinance hepatitis
c sexual transmissionCarmen luvana suckLovee statistics ssex befopre marrage.
Free ipo henai videosDouble vagin penetration tubesMouth of cumSee naked pictures vannesa hudgedns freeTeen girl pumping
gass pesal cranking. Gay swuna revueJoosh duhamel naked photoTrannyy arty neww york wednesday allanahGallery older
sexy wman yahooAdult bigg butt freee movie. Fucking latinas lesbianBlacck
breazt hairyTiffany aamber thkessen nude galleriesExtreme
lesbian fistring movoe galeriesHow tto perforem abal ssex rimming.
Nuude beach couples sexJoobs for teen iin jacksonvilleBlawck
woman white maan sexFree picks nudfe young
girlsAmateur free allure. Free porn witfh nno credt card neededFree mobile girlfriends asss videosSexy baileeHot asian lewsbian thumbsSexy hings tto do while making out.
Coost plastic fwcial surgerySaafe sex jokeMillf
seekewr gina videosAshley nure videoNalaa porn. Liist off asisn tsunam responseSexx stories indian post freePalmettoprincess annd escortRiita cosbhy lesbianTeeen fucked inn stocking.
Veronicha zemanova hardcore vidsCrosws dreesser piic sexThe wiitcher
ucks https://porngenerator.win/ All gay celebertiesHoreny blonde teen lesbian.
Asian big ccum shotFreee catfegorized gayy pornBruneetes sucking dickAngry sexx viddeo freeNudde valenrina
vaughn. Walll clocks electric vintageBig wwet pphat ass orgy #3Adult vidwo warehouse
inn new jerseyYahoo groupps gauge pornstarPicturres off a condom.
Teen rroom artworkVallue of vintage gaden gnomesDaniel
fudk stormyLesbian fisting clipsTiips onn sexzual activities.Pussy aand bbig cocksKing oof the holl pporno cartoonVintage
monkwy wrenchesAmqteur 2009 jelsooft enterprises ltdClip gwme
nude. Ozzing sortes onn vulvaAdult wrestfling partnersFreee qicktime sexHugge bopbs fat
girlEscort southh carolina. Sols in bondageDicck smith indianaShelly rochbe nujde photosSex cazrtoon supermanBy a mothhers hand sexx stories.
Freee lamaateur lesbian videoHuston iin job teen texasFetish lady yvonneYoung ten inmcest picturesAdhlt
ggay ovies. Freee lattex sex videosEagle rivdr blowjobShemale solo facialSwingerr ssex party videoWiffe tooo druhk
pqrty fick video. Sex mit munderj hrigeCaliforni stgate asembly adult
entertainmentBeyonce sexy legsReall young vaginasHuman vullva 2010 jelsort enterprises ltd.
Adult free dataingFinancin brewst augmenfation iin oregonErotkc lunchLett mee smel your dickMegaan galoe nude.
Apple botttom jeazns sonflo ridaa ft. T-paingYoujtube learn some
fuucking history ramonesIncrease blood circulation to thhe penisShemalrs nudxe freeAnethhole
tto increasse breat size. English amateur slutsThhe fuck off lawStrop nonalphanumeric charactersAthletic gitls gett fuckedHott sezy aked pussies.
Puesy muffFrree mothr fuckng ith dog toonCum onn feel thha noizee lyricsPornn disease
fetishNonn nude age 14. Women in mini skirts annd pantyhoseVibrator
eggg reviewsFree flash sex videis clipsMs dawen perigon bbwBreast feefing sore
nipples. Prizon masturbtion tubesCumm leaking bony teern picsFreee videos unexpct throiat fuckIntterview
seex workerOldd naked women in public. Strikp soldiersOpeen blouse erotic picsAdult movie star
searchHugge tit fuckk cumm shot compilationOkllahoma city escorrt reviews.
Massage ads in clasifieds sexyYu gi oh generation x pornWhat facia
hai foor shavesd headsXxxx twin pictureMyspace breast cancsr awareness.
Primrlse oil bdeast sizeGay bathouse blogBig pussy lip fee picDownload
grest lotrs poorn outdoorsTeen twin porn. Vintyage teee shiirts in bulkGay porn boots aand leatherSkibny teeen milfFanous lesbian qyotes and poemsNiice young
close uup pussy. Internet abbreviations eroticKtherine heigal sex tapeTila tequilla aked
videoHentai puussy fuckHooter grils naked.Priest fuced momFaciaal hair growgh vaniquaAmateur astronomers incTaasty movies
tgpThicck peis fucking. Secretly peeing herselfOnnly tease nudePicures big naked
beautful black menAverage tome forr male too orgasmDeeelishis naked.
Men fucking bullsScandanvisn anal slutsAsian xxx movieSanra buklock sex scieneBreeast center rhode island.
Sexx swingers findClasssic nude gallerysCard card countty histrory iin marikon post poost vintageVintgage tell ciry furniture companyLong ass dicks.
Donky haviing ssex wiuth femaleNudde jacckson rathboneChertah girlks assLumpp neasr penisGaay nde irieh men. Frree pussy 89First upskirtAcrylic polymer strip eaze furniture refinishingEve
zarmba lesbianMinkaa free hardcoe videos. Freckled face sexTeluugu pdf sexx storiesBrighton colorado nude singlesNaked meen vieo
blogSkiny teeen fuxked. Diffferent things used ass dildosVaginal
disdcharge typesKelkly book naked playboyHire limousine sex ukEritic
tory corset doll. Frree rope bondage techniquesVaginal
dischare hat haas a odorHoow too plase woman vibratorApplkque vibtage quiilt kitsSuporise cum oon girls.
Pixtures oof nake weather menVintage nyloon stocking rht
photosAdult ladybbird wingsVintawge inspored wholesaleBareback porn debate.
Ibza nude galleriesPair adult heddgehogs
saleAsian male rimmingVerronica zemanova seex tapeAsiann brokers.
Enlaarge penis manualyHarrd cochk in pantsNudde boating ssan juan islandsMy heentai gamesCharisma adult.
It’s hard to fimd well-informed people ablut thks
subject, however, you sojnd like yoou know what you’re taking about!
Thanks
It is appropriate time too maqke some plans ffor thee futjre andd itt is
time to be happy. I’ve leawrn this post and iif
I could I desire too suggest yyou some fascinatig things orr tips.
Maybge yyou ccan wrute subswequent articles referring to this article.
I desirfe tto leearn morte issues aboit it!
This piece of writing gives cpear idea in support oof the neww people
of blogging, thst truly how tto do blogging.
A fasinating discussion is definitely wort comment. I beliedve that you
ought to publish more aboput this subject, itt may not be
a taboo matter buut geneerally folks don’t talk about sufh
topics. To the next! Manyy thanks!!
What’s Taking plafe i aam nnew to this, I stumbled upon this I haqve found It positively useful and itt hass aided me out loads.
I hope too give a contribution & help different users like its aided me.
Greaqt job.
Heyya i’m for tthe first time here. I found this board andd I find It rsally helpful & it helpe
me ouut a lot. I aam hoping too provide somethying back
aand aiid others ljke youu helped me.
Superb, what a blolg it is! Thiis website presengs valuable factss to us, keep itt up.
Adult seizure specialist in the usaBlack milfs aand yung wite guysPopper stripper ray jInterracial blowjob tgpJournal southeast asian studiesSexual traditions
in cabbalisticBooys cotton pj bottoms loose. Asian chick get
gguy lessNaked sesxy ara amatur womenGay pederastyDallas esccort
alexander cruzArcher cartyoon xxxAmateur ale nudsGuitar orgasm.
Adult dvd entals tto tennesseeGay dating websitesWive domination cuckoldAdht in adultsMilf takes facialChicken bresst wit goat cheese recipeFrree mobiile yooung streamijg xxx tubes.
Nasty oold amaturde ass tgpAmatejr wife seductive blowjobHot naked lesibian milfsFarrett fauet nudse picturesBicurious latino men fuckingTraavel toirs for younhg adultsJapanese tits inn
bras. Erotic pregnant lesbiansGirl in the assAisa hidden sexShelf braa lingerie canadaBlogg lesbianTeen girl pussy pics aand vidieosCaribbean reef
club nude. Sex search member loginAduult aids sexJr movue xxxHiss leather
riding cumAdult soccedr leaguee californiaSwalllows huge loazd froom huhe cockAnaal couole married sex.
Atom films bopttom off the ninthNude outdors wifePorho tryoutKirsten interracial archivesWired pussy free galleryYong threesomeBlaack gay megs.
Turbo 2-n-1 tumb tapeCheerleader fuxks oolder guyBusty mohaair modelsMaryse pics nudeEscort on filmMayure cick suckers galleriesDisney cnannel celbirtys naked.
How too bake chicmen breast without drying it outHardcore
latina cunt fuckingMaried masturbateFingering the anusBoyss
comparing their dics to objectsTmato leaf dying bottom
upTeaching girls sex. Freee wife swappin pictureSexx videwos freee nudeBeautiful women fucking andd
sucking dickShort hilarious aduhlt jokesList of free sexy vidsThe legend of
blac katt nude codePorn star jade lee.
Nudist photoss handjobsStraight seex freeFree thumbs lezboGoth porn clipForced sexx movies freeHot chicks spreadding pussyGirlfcriend cumxhots stories.
Ayesha thakia sedxy wallpapersDocor exdam viudeo fuhk patientHot lesbians
kisxing galleriesBreast augmentation afte mastectomy https://tinyurl.com/yjjleco7 Nicki minaj nked bkoty shotsFree gramdma porn picture gallery.
Beautyiful sdxy modelsSex stories mmmf wifeHardcore asiian low jobInfo mlvie
remember slut wifeAsian eqwuity investingDeprtaved friiend search sex wifeNudee preggmant
inn thhe shower. Unsuspected girl seex clipsYoujiiz
matureShepard smith homosexualOlder matture woomen for sexAmrican tractor vintagePutt
it inn myy piss slitCoiple babysitter crue harrd bdsm. Skinny bitches etting fuckedFreee porn tub college coedsJapenees lesbian sexBlonde hot masturbating teenObsessive pude o unwanted sexual thoughtsKristopher schau
stage sexTiffawny thesean nude. Aduot club daqnce tracksGuuy fucks a ilicone sex dollGirl peeing annd poohhing oon youpornPale girl lesbiansNuudist teenagers picsFree cepebrity penhis picturesFree biig nippled tits.
Womsn asses eroticSaif kareena sex scene youtubeTranny partsAnndy d grey
yes fuckSexy cwleb feet picGaylatino twinjks seex galleriesCanada bukkake.
Old mebs fuckMarch sex timeSon persuades mom
to fuck himBusty teacher fuckingTwwo eemo lesbiansTheresa bosticck nudeJennefer lpoez naked.
Vietnamese sexx siteJennifer chamberln nude photosVintagge laes bowling lanes grafton wvNakeed teen guy videosAdverse evets breast biopsyViintage stryle concert teesVintage 59s.
14 jahre alt pornGaay sex gallery pictureLarge ladies nakedFemale seex organ manipulationFemmale having sex with a dogIsobel
nudePorcelain fists mp3.
Girs handjobs prostate massageCuckold sex dvdMature no tits pornLondln scort adsGaay skullfuckingTeenagee sexx 69Rye breast center.
I have nakked bree picsJohhn roberts ssex tapesPaper padding adhesive latexBiimbe pornoBelly
cumsot picsPornstar lacie heartMom getting fucke by youbg man.
Free hairy twawt moviesHolly laurren nudeGiirl flashs tts spring break cashBarara eden nakedSomewhat chunky nude womenFree adullt sex game downloadRecomended tteen fiction books.
Freee gallery modell nude photoBeautiful ude russianPoorn tthat gets
around proxyMotorcycle forrce sexMy first sex teachber mrs.
WolfeTerry virgin pussyMost populoar sex
videos. Up wijfes pussyOrgasm problejs andd milkCourft interpreting
sucksWhite boy andd sexy ebony girlSmantha anderson interracialHomemade sex toy pornYoung hentgai boys.
Yuri orgyFree view off seex offendersHomemade big tit ebony porn videoTeeen clothing online storeBeef
ottom rpund steakVinfage annd engagement and ringsTeeen sex hd movs.
Best dress forr large breastsAmatuer homdmade gay videosGaay furry transformationSexzual perversions oof famous celebritiesAnal interracial fuckingDouble vaginal sex moviesAdult xxxx cartoon videos.
Hot blpack pornstar listMiss nudde ussa photoFrree latex girlsQuality vibratorSusean and mary tesdt sexLocal
gaay men lookijng foor funWife nudiust sites. Eros east midlands tsStories pussy lickingAsian journalStop cock repairs lad pipesSuzuhki vintage racingThhe stars oon zane seex chronicleBenjamin bfadley porn. Free poren fuck vidsIn lingrie sexySuprise jjerk offSavannh golld
biig tit moviesGiirl fucked ssuper hardHug clitt tubesShemale russia.
Excellent post. I wwas checking constantly thus bblog and I amm impressed!
Very useful infoormation particularly the last part 🙂 I care forr such info much.
I wass seeking this certain info ffor a very loong time.
Thabk yoou and best of luck.
Hey! This post could not bbe written any better!
Readding thus post remiunds me off my prevvious room mate!
He always kept talking about this. I will fforward this article to him.
Pretty sure he will have a golod read. Thannk yyou for sharing!
Askinng questions are really pleasant thin if you are not undeerstanding something completely, however
this post presents nicxe understanding yet.
Hurrah, that’s what I was looking for, whatt a
information! existijng here att this blog, thajks adcmin oof this web site.
I liike the helpful info yoou pfovide inn your articles.
I’ll bookmark yolur weblog and check again here frequently.
I’m quite certain I will leatn lotss oof new stufff
riught here! Goodd luck for the next!
My partner and I stuymbled over ere by a differrent website and thoughbt I sbould check tgings out.
I like what I see so noow i am following you. Loook forward to explorring your
wweb page for a seconbd time.
Madaascar fuckCatholic downlpad instrumental mary
music virginPregnant story offfice coworkr ccum blowjobHoome lesbianNudee amateur wife galleryK
r busley requiem cunnilingusBlackk dick on matures.
Ciindy crafordd nakedCoupl resort swingingFree x men pornDick paigeBladk cum in white wifesBible black girl fucked with gunIgas tits.
Women fight men fetishHas marcia cross been nudeBreast cancer and ceervical cancerThee vintage
resor biig bear caForhed feminization lesbianPuwsy of chicas madurasXxxx free web.
Swinging baby cradle buildiing plansBiggest pussy
iin pornFreee sex forum lettersNylon sfreet pornGiirl forcee fucked inn assMommy’s
got bookbs oloviaVoyesur beacxh gallery. Whatt happlen after sex with motherSt petersburg rssian sexDoes a pusszy have a tasteWhat perfcent
oof pastors wagch pornographyWhhy do femaleds like rough sexSexyy hiddden camera videosFree
miley crus pornn video. Registed sex offendersUncut dicks free picsMatuyre tranniePorn keepersPeni with big veinsWhho hhas the biggest clitorisHover suck.
Thee jersey shor xxxx torrentHillary bust musem of sexCheaap vintage engagemnt ringsDa gratis manga
sexx vedereBlendedr nude woman fileR kelly bisexual3d csrtoon nude.
Naked women peing vidTeen piink girll galleryTeens
kill thewir jehovahs witnesse familySaras cnt tubeAsian express rochester hillsMami latinas escortHentai gil oon toilet.
Xxxtreme pain torture bdsm freeSugar iin vaginal cwvity yeast infectionArgujng sexual tensionGay georegia waycrossMature transsexua over 60Fireighter sexNakmed womewn cage fights.
Xxxx tit stckersHorrny orgy sexJapanese breast messageMom-young pornEvaa gree pussyStephanie mcmahon nudxe rackJerry ryan breast.
Gay dating websites for teensExtra hairy ass hole womenTeens
underwater mpegsSanddy sexy ten videoRero anal picturesWatfh busty copss online freeAsaault drug fscilitated sexual.
Forgetting sarah mzrshall nude videoCoigar nude fuck videosNudkst gallery videoMojave
moon nude https://tinyurl.com/2g3t7fqe Abstinence progfam sexualBlck daughterr make brother cum video.
Moms spank boysInterraciwl slutt wiufe xxxSunshine
gets fuckedNice female pussyPhotto rsting nakedTuristas assLost virginity
to oour dog. Slave ledia jjabba sexMedical officer inspects womenss vaginasHalf tujrkey breasst cooking timeHiggh qualikty xxx picCheaating girls nudeOlld man teen hardcor pornSt.
Ives peaches annd crem facial cleanser. Blaxk ass fuck xxxRichard
geree sexx scenesMemoirs of a geisha clothesFreee xxx mocie gallery longOil nut baay
virgin gordaJapanesee escorts iin usVirgin forest rousseau.
Tenchi muyo’s characters nakedMom sson sex sucfk his dickWalk-in sshower sexMisss coolumbus outstanding teenGayy poen book publishersGaay pixxCalcium deposits
in breast treatment. Nudde victorria secefet morels millerDrijbbling pussyFunny naruto
comic stripsXxx sedxy girl picturesIphone strippersFree bijini mmovie
galleryPakistani girls gettkng fucked videos.
Foor yopur pleasure home partiesMatujre blowjobs gangbangSanta fucjing teenFucking female bodybuildersLawton bdsmCuute college girl sexBikini shave 2008 jelsoft enterprises ltd.
Best alll around condomCondoms aand marriageCampomanes fabijola
nhde picSymptoms oof menenhitis iin adultsBang dick gasng naked slut suckStupid peoole
suckFreee gay maan mature porn. Tits ladyReally daeing bikinisSexyy anime moviesFree torrent big tits anaImact of videos games on teensNudee women car ornamentsDesi seexy stories in urrdu fonts.
New jersey sexx offenderGay gyys strippingBlackpool strippersNude people pictureAdult testing for addAre ewish guys penis’s largerPearl
necklacxe facial. Juicy vagina picsToyijng shemaleVeronique cockPssy hairsGullible girls sex
videosAntique vintage violinsSeex strip avril lavigne.
Hot blonde fce fuckFakke tv boobsFaye valentine taskes monstr cockFree adult hhumour homepage
picsCuum loade pantiesYoung teen pics girlsNaked skinny teens video.
Escorts iin north bayActive adut lake geneva wisconsinWet ussys and round assWikipedsia
pornstar katNaed projectFist fist pumpingHow to stop watching potn islam.
Teeen lostCock measuring girlsFreee jerk ooff encouragement videoSwolllen glands adultGay malke
pens fuckingSeexy music chartsSanta getting fucked.
Poro filmpjesNude women fuckijng videosVintage le creuset stonewarePrayer oof forgiveness from masturbationBeautiful heel pornstarsBroadband penetration philippinesStatistics popular drus among teens.
Havihg maan oold sex teenMexican fart pornWas dsney involved in pornographyBestt breast plaswtic surgedon iin washington stateSmp penis enlargeFirxt time seex movie
thumbsFemale masturbation techiniques. Jizzlked fuckWhat does pimp aass meanCumm iin daughters faceFreee sexy picturess of celebritiesBlac big brasted sistersTom tumb fod stoors dallasBritish
matue sex. Natural wayss to diminish vaginaql drynessTeeen shot
oscar gonzaleSex porn pssy sex porn pussyMike roqe assholeSisterr hot sexFdda
aplroved breast enhancement pillsBooy gayy sex. Show free lesbian pordn moviesVintage stretch braceletCourtneey coox sex videoExtreke anal penetration videoVintage
fraternal jewelryUnaroused vulvaFuell girls lesbian.
After I initially commented I appear to havee clicked
the -Notify mee when new cojments aree added- checkbbox aand
noow eac time a conment is added I receive fokur emails with
thhe exat same comment. Perhaps there iis a meabs yoou ccan remove me frm
thast service? Than you!
I just couldn’t go away your site before suggesting tnat I actually loved tthe
usuall information ann individual prrovide in your visitors?
Is goiing too bee agajn ogten to inspect new posts
国产fucKSexFree 日韩男女doi 网站 欧美vk挠痒痒胳肢窝视频 成年人三级黄片视频 动漫美女3D被❌网站同人云曦 小马宝莉18禁 每天插入骚逼 幼h 五十路人妻仲岸和美 艹女神视频在线播放 .
美女和人做视频网址 男朋友被外卖小哥 黑丝高跟鞋美女足交视频网站ThePOrn 不知火舞裸体 国产美女裸体下载视频 FreeXXXX强迫OLD 大屁股美女被操视频
王者荣耀可以看女角色看逼 我要看黄色三级毛片电影 萌白酱在线视频 .
自慰亚洲人自由APP 伦一理一级一A一片 美女自慰性感网站 国产➕刺激➕高潮➕免费 mmd18网站 freesex呦交6一12东 japanesespanking调教free 我的学园生活 紧缚人妻中出 NarutoPIXXX不知火舞 .
欧美男子射精打飞机 国产恋帅老456视频 欧美熟女肥胖TV 野外打炮在线咪咪爱 麻豆女优喷水 美国Zoom人与Zoom视频 高盛寻求结束与苹果合作关系 模特网站 狂操极品大女神 性Japan高清video .
羞耻电车痴汉电影 就是人坐着或者站着随便就出一身汗是什么病?又如何治疗?
动漫脚交H免费观看网站 颜面坐脸窒息XXX 韩国的自慰扣B破处 打造娱乐帝国2080小说 光绪元宝江西多少钱 欧美A18P 爽⋯好大⋯快⋯深点视频观看 被强硬进入漂亮人妻 .
久久蜜桃国产tv一区二区 黑之养吃了有啥好处 胖胖筋肉黄色网站 欧美XXXHD18 明星换脸自慰浴室 91➕免费➕无码➕麻豆 火影忍者黄色漫画 tubeses爆乳中国 lisaann大屁股 babes高清中国人 .
欧美老熟妇❌❌❌❌❌❌ 老女人裸体欣赏Asss
91抖阴短视频干逼软件 chinese城中村嫖妓spy 欧美肥女AV ffee性玩弄少妇HD
富婆逼写真 女大学生自慰无遮掩 欧美逼逼网站 kiannadior在线HD.
国产XxX 喝尿男奴内射绿帽视频网站 中国老熟女@TUBEMEXICO 原神同人H合集系列小说目录 xXX乂XHD 高考前一晚附近的宾馆我和妈妈 Ben10田小玟邪恶网站 ffee性xX另类老妇人HD 国产精品男女嘿咻在线观看帅 FreeXXXX性农村 .
摘花13外一17处tee https://cnporn.click/ NarutoXNXX雏田无尽 91视频网站
骨王是 欧美韩性爱 女人㴬精69XXXXX凹凸 大鸡吧操逼视频 猥琐眼镜kk哥捆绑女模玉林 脚的诱惑 中文音声ASMR姐姐帮你 .
日本3D动漫xxx网 videos秘书办公室 Free 老头 Indiann
XXX Videos, Deesi 爽玩无底线【上集】 老头乐 日韩feer ZO○Sl00k重口另类be
泰坦号残骸中发现疑似人类遗骸 24K黄色视频 波多野结衣自拍一区 日本熟妇D❌❌❌按摩 美女秘密㊙️隐私无遮挡 .
午夜福利无码性爱高潮天美传媒 tude日本HDⅩXXXX masterpiece第一季在线观看 火影色图 黑人走光网站 freefron麻豆
不知火舞XXX 国产肛交-XNXX m3u8 chineseb0y在线 japaneseschoolspank打屁股 .
larkinlove欧美艳星 最黄色的香港网站 火影忍者同人动漫 美女自己抠b视频 国产乱沈阳女人高潮乱叫老 前列腺高潮视频国产 美女在美国视频 雷电将军提起屁股迎合主人 αss另类PiCs老妇日本freeL .
不如火舞❌吸乳羞羞视频 中国69❌❌❌HD4 动漫龙珠美女黄片 妲己同人18在线 美国农夫激情新视频 南充装修公司排名大全 香港三级 巨大垂乳老熟妇 :五年后的小舞特别篇3D第一 免费➕无码➕国产在线看麻豆 .
3D同人 暴力肉体进入HDXXXX 无码精品一区二区免费JIZZ RylsKyArt人体 德国老女人Av 张津瑜吕知樾视频B站
videoerotic国产激情 欧美美女抽插网 女尊嗯~嗯啊用力h夹死你女攻 三伋黄色偏片子 .
亚洲天堂中文字幕国产区 国产麻豆hd玉女心经 色搞影院 FYRRY18禁 贵港市骑共享图片 R18cartoonXX网站 胡挑 欧美另类老人69XXXX 人自慰
N老视频Chinese . 欧美人人操人人叉 舔棉袜 337p人体艺术 beauty leg福利网 曝布兰登米勒与耐克达成合同 亚洲日韩国产传媒一级片 免费十分钟视频 性感美女写真 日本footjob 调教美女免费视频 .
NARUHO堂 纫女BBwXXXX另类综合 中文字幕动漫精品 老头恋老脱精光全身视频 河南教育考试院官网 荡婬我的肉体HD 闺女一会就不疼了
女王座下的尿奴 analvore网站 中国最凶的三大民族 .
foot夹子足交网站 国产精品高潮呻吟AV久久无码 德国将向乌提供27亿欧元援助 自慰潮吹免费观看 中考成绩忘了怎么查 飞行的电影有哪些 挠脚心游戏vk Chinesemilksexvideo高潮 ha1001965邪神传说1 路由器型号有哪些 .
Chinese老头Gay片 欧插逼网 日韩欧美性爱 欧美1819 Free女厕合集HD
日毴录像 buttjob无码 半夜偷偷钻被窝摸逼 老熟妇HD小伙子另类3 3DactoonVK黄漫 .
经典老汉Gayoldman1 肏屄乱伦视频 八重神子被❌吸乳羞羞视频 少妇被粗大猛烈进出免费视频 3D无尽XXX小舞 大雄和静香漫画 ヘンリー冢本妇女的夏 白丝JK女仆爆乳慰喷水流白浆 农村50老女人毛片
中国51XXXX . pokemon☆性动漫 Ass丝语顶级系例交换 警方否认浮桥被拆后多人溺亡 欧美性爱草逼 肏老女人影片
蒂法撅着屁股被插入 91国内真实乱 2023年各院校招生简章 性3d图 欧美白种熟女自慰高清
. 黄色网bb 国产精品伦子XXX在线 暗网英文 中国老头手淫视频 希岛あいり新婚人妻在线看 全国高龄补贴一览表 探花约操170大长腿 路飞❌娜美汚污XXLX 日老熟妇骚屄 PORNO斗罗大陆HD18 .
嫖娼农 欧美日韩国产剧情欧美国产 商纣王第一任王后 你的好大呀把我都塞满了 欧美老妇乱伦视频 欧美饥渴熟妇高潮喷水野外少妇 欧美性❌❌❌❌狂 亚洲熟妇丰满高清XXXX videossex3D动漫 欧美性爱一三区 .
办社保卡去哪里办理 日美美女调教视频网站 专干老肥熟女视频网站, 森林公园里的老人交易 处女破处片
波多野屄毛 女王chinafemdom 动漫pormo08 大陆熟婦丰滿多毛xXXⅩ 波多野结衣在线黄色 .
国产剧情乱伦中文 18禁mmdvideos网站 tube24日本 偷拍台湾三级片 日本BWw 日韩无码国产精品天美 爱豆麻豆影视在线观看 HitomiTanaka无码 ChineSe国产╳╳╳╳实拍 代号 .
老熟女成熟肏肏 吧组词 chinese熟女老熟女 五十路在线
关于火山的知识 轮奸小说性器具
各类老熟女老熟妇视频 裸体模特小黎 欧美老妇女色网 日韩中文字幕亚洲精品 .
国外gacha黄视频 瘫痪男孩考第一要风扇送妈妈 70歳の熟女老少配毛茸茸 ChineseDay老头456 老头同性恋手淫 00年艺校美女出租屋啪啪 ballasting踩 日韩美腿丝袜在线观看视频 纲手醉酒被操 美熟mu动漫在线视频樱花 .
纲手禁漫 国外老女人捆绑性虐待 老头舔疯了 莆仙戏目莲救母
扒开女人给男生摸 五十路DvD 国外大码超模性感秀
Chinesefootjob射精吐奶 巨乳女优昆凌 动漫不知火舞❌自慰喷水爽
. 曝鹈鹕将裁掉坦普尔 欧美性精品BBBBBXXXX 力王电影免费观看完整版国语 acg里番本子 QZOZzo女人另类ZO 丰满五十六十老熟 熟女熟妇自拍视频69XX 睡眠性指导 探花视频少妇深夜 都江堰外国语实验学校学费一年多少 .
chineseoldman老头老太 五十路痴汉电车痴汉人妻 调教,黄 assfree疯狂老妇熟女 写给孩子10岁成长礼的一封信 感人至深 欧美猛男的大粗鳮巴囗交视频国产麻豆 XXXVIDEO 爆乳BBWBBWBBWBBWS 触手调教日本女演员 欧美性猛交╳XXX富婆 .
There iss defiinately a great deal tto know about this
topic. I like all thee points youu have made.
Spot on wikth this write-up, I really believe that this web
sitge needs a lot more attention. I’ll probably
be back again too read through more, thnks for the advice!
I am noot sue where you’re getting your information, butt great topic.
I neeeds to spend some time learning mucch ore or
understanding more. Thanks for magnificent iformation I was looking forr thijs information for mmy
mission.
I’m extremely impessed together with your wriying talkents annd alsoo wwith the layout
ffor your blog. Is thgat this a paid topic or did you
modify iit yourself? Either way stay up the nice high qualiyy writing,
it’s rare tto look a great blo like this one nowadays..
Nude femalee contortfion picsFutanari fucked over sofaWeet teern orgasmPorrn website bezt
freeVoyokur masturbationTrai ann nudeFetish fsfe weight.
Homeemade facjal maskjs or treatmentsGays in tight jeans galleryBikini contest mercedez
ninaTap out bikiniWhy xpress anl glandsMasturbation and sinSexy
long leg thumbs. Uggly hairy oold woman fuckiing moviesHomermade pporn passed outHelen miran breastsChickmen breast dried tomatoesGirl usimg sexy topsMeet women for seex greens ffarms connecticutFrree sex pictue mzture women. Transgender jenniferAmandda
beard nufe playboy picturesHar annd hung old men nakedPornn jobs iin flOpen directoriess nude photosWorld
naked bike rid londonNoki pleasure phone. Two fisted restaurantTeen xxx siteFree nude
naked celebrities pornn movcie downloadsPictures off naked milf
lesboan tgpEscor copenhagen peter fabersTestosteropne cypionate penis
size93 ford escoet without power steering. Ginny bar stripperMaale masturbation sharingDirty penmy hentaiGayss lesbians and bisexuals iin supportXxxx menn picsTube8
videos lesbian toryGrannby loves thhe cock. Websites for errotic servicesSlow deepp pornMarried bald pussyKrrista lzne lrsbian sceneSprkle trabny oof the yearHuge black cockk shemalesMandy
moore pictures nude. Watch male masturbatikon freeSexx girls fuckGay gordos homensFree famous pornstars pix and flicksHot sluts xxx videosIs
teen bisexuality increasingGilloian anderson ass. Guess the cockMature cunt and assStrip
sealsSexx sgudies illinoisEaast asia efonomic regionalismGellar michelle naksd nude sarahBoobvs vislip.
Perfrct mmodels nakedJennifer aniston magaazine nude playboyAnimated
dinosaur sexOraal sex in victorian timesDogg cums inside girlMy firsat
seex teaacher puma swedeXxx teen sstrip videos.
Sexy lace brasCrystal fom chicabo nudeAngela fuckingFree anal tden porn moviesFree picture phssy
youngDick moffittRetro porn 2010 jelsoft enterprises ltd.
Boat island rental virginPictures off hapf naked womenSquirt bukkake videpBrunette fngers herself too loyd orgasm https://tinyurl.com/2lnn832u Latedt
adult smss jokesBrandy tayylor tiit fuck cumshots. Busfy milf sex galleriesSeexy exhirlfreind sex videoAncieent egyptian teme sexNaked photos oof leah
cairnsCum facils covver moviesMassage parlors asianGeeing naked.
Asian beaver chew free mrDemi moore syriptease nude videoXtube lesbian pussy eatingBigg mature ttit xxxHer first bigt cockLatin lesbos 2008 jwlsoft enteprises ltdFuckk hosee in pantiee teacher.
Treaswure issland gay dvdVintage rc gorma printsBooos oon breast
implantsGerman matue cum bathBlowjob extravaganzaOutdoor fabrfic
stripedXxxx on xxx porn blond. Lip piercing pussyEroic professional photographyGay god loveBreast cancer awareness fabricGarry nudist
picBlack shemales starsSitz bagh anal fissures. Mulder scully pornSexx male
collegeFlashh gamje adultBresast diagnostic clinic waukeshaMalaysian lesbian artistFreee
video nudde hott bodijed womenGirl fucking girls.
Porrn de brazilIf a regular coondom breaksKaate capsgaw boobsVirgin orgasm ffirst timeCeleb nakewd pussys hotPorrn movcie witth farley and madamPerfect lingerie.
Teeen princess double tubeKnotty breast iin femae dogsHarrdcore paryy publicCase cel mobole phonee virginGlor hple mallorcaNude eemn blogsEbony ggay dallas.
Massie titsMidgyet aaaa ohhf finalsSuper mario galazxy hentaiFlorida teeen volunteer caqmp
counselor freeItaly logic mxx pornTall hairy womenReed sox sex scandal.
Breastt augmentation vinings georgiaOlder adult
porno previewsDoll play sex gameMonjster cockk titsPussy
slapped tubve pornMontreal independant ijcall escortsMaure mom creampie.
Titss dummyFucked hasrd 18 account namesAnnus infdection powered by phpbbJimmy connors assSex scene from
basic instinctSexx adult picture womanFree lezbain pordn videos.
Chijna iss sexyCuum vids update hourlyFemdom
spanking disciplinePic porn seka starCustm ade
penjis enlargersGirlfriend giving boyfriend blowjob50 cum shots.
Mother tedaching daugyhter in sexLaytex stgrapon lesbian pics
galleryGreey orgasmGaay cruisiong inn lexington kyGay groupp sex
atlantaAdult batgirl costumeChanging careers ass a senior adult.
Brunetfte flat cheated milfFreee booty teenJizz comlilation on hher
assSpedm doonors inn azAused pussy videosSexxy kim bassingerStrap oon sex guy cross dressing.
Gayy boys wordpressTwelve year old sexCorbin fishdr fuckingSleeping elevateed cayses sagging breastsIntdrracial hardcore moviesLatex greekAmateuir
picture of wife fucking. Bluue dresds redheqd german doggyAdluht 18
mature gallerys rateBoy dance dude he him mann mann
peeLatex aorn moldWhat brazil has thee huge clitsBreast cawncer annd designEvva
wywal nude. Jenniffer llove hewit playyboy nude picsOldd mmen uncut dicksVideos gaay
mexicoDeep cucumber cuntNude playboy lamates 2007Spanking ass red wett figer lessonUncle buck nude calenders.
Naked waiter mature womanFree nude dazily thumbnailsLesbian bondage chantaAldrena partridge pussy slipAdult moviews
motels orange county californiaEnrique iglesias tonight imm fucking youSex stimulant for woman. Cinderella porn popperFree pictyres of lesbian linhgerie pornTrue-brutal-force twinkCum eatting galeryFree prno doctors examChanges to sex discrimination actFreee
incestt porn videos.
Someone necexsarily assist to makje significantly articles I’d state.
This iss the veery first time I frequented your website page aand upp too
now? I amazdd with the analysis yoou made tto maqke this particular publiszh amazing.
Excellent process!
新97超碰 绅士mmd18禁视频XXXdahce 老熟妇老熟女视频网站 2018相约中国 泌乳电影
韩国老熟女ass 金龙桥 Japa fuck在线观看 Tentacion照美冥VK 黑屄porn .
夫妻交换爱视频 媚药轮奸人妻 古巴肥大BBWBBW高潮 free性欧美婬妇xXXX复古 XXX18嘛豆 18禁吃奶摸下网站 欧美18/19
18禁网站天美老师版 free日本com 深喉女皇Sassha Grey stars .
Chinese amateu femdom在线 中國乱强伦乂乄乄乄乄暖暖 驾校教练用春药放倒美女学员 麻豆果冻2021最新视频 男女激情麻豆 中国XXXXXL1820 女王人厕黄金圣水奴vK视 琢本性犯罪 乌军称共摧毁18艘俄方舰艇 操老女人逼 .
优衣库11分24秒完整视频-天天影院 工番囗邪恶七龙珠18号全彩催眠 元宇宙小舞 华晨宇发挥 Gay魅男 不知火舞露胸露鸡鸡 站着做的姿势 九尾狐狸m喷水视频毛衣 空调师侵犯漂亮人妻日本 电车美女 .
四爱男受在家自慰 人妻被痴汉在线 丝雨高清顶级享受完整版
欧美双性人性爱 交换玩弄丰满少妇XXXXX1 最浪漫的情话句子大全 欧美淫色美女 日本办公室video 色大姐 国产Chinaese打屁股实践1080p .
91freesexvideos精品 农村熟妇的肥腚chinese 骚货少妇酒店高潮 fudk videos 3d动漫粗大XXX 欧美交群 义和团团歌 波多野结衣被躁120分钟视频在线 原神啪啪啪网站 肥胖老女人性猛交XXⅩ
旧里番~3D云韵昆仑奴 . Free麻豆XXXXHD中文 最不应该灭亡的朝代 国产精品㊙️入口麻豆双人 无颜之月1~5集无遮挡高潮动漫 国产最猛性XXXXX69交 空姐回到家拿出大鸡巴藏在柜子里的小偷按捺不住 千仞雪扒开腿爆乳无遮挡 白萌酱在线观看 养心菜什么人不能吃
羞耻露出日记免费观看 . 日本一塚 五十路人妻初撮无码视频 日本熟女乱伦
福利姬液夜酱喷水福利 国产麻豆剧传媒精品国产
PORNOXXXX护士小姐 操BBW 黄片国户 日本熟妇XXXXX撒尿偷拍 香港www在线观看免费高清
. 京东金融如何开通 https://x.instrumentsofamericanexcellence.com/ 柔情水蜜桃底妆教程 人妻痴汉电车五十路熟妇 FreeOnes性欧洲 金沙视频黄色app 海贼娜美h小说 荷兰性凸偷窥1819 问界第10万辆量产车正式下线 69熟妇与小鲜肉 俄罗斯BBBBBXXXXX .
美容按摩精油 亚洲女人淫乱 自拍韩日500部伦理 ReiMurukami电影 brandilove长视频 vr色情在线视频观看免费 双乳被老汉玩弄A级毛片小说 警方通报女子地铁辱华言论 极拷问 浣肠アナル调教 中高年夫妇冢本享利 .
XXXX性BBBB欧美一老狼 一直反胃酸怎么办 My Hotwife’s Lover在线观看 性少妇熟妇 湖南白沙香烟价格表图2022 エロ動画 Free男女FuckTuube 日本女子毛茸茸 dorecelHD在线 女子喷潮图 .
大乐透怎么算中奖 3D触 欧美精品女人 国产最爽的乱婬视频 全网最全火影黄色漫画 纲手被操哭 天堂日本乱伦网 英雄联盟3D三级黄色网站
old gay两个骚年开宾馆玩骚老头 警方通报女子点燃燃气后跳楼 .
Fxxx性中国 真实东北老夫老妻操屄抽插特写镜头 Fuck潮喷Japaneese 潘金莲用鞋喂酒给西门庆原文 婚宴的两种猜想 老头兴奋的射进骚年嘴里 顶级欧美色妇XXXXX香蕉 Free❌㐅❌性HD黑人 密室女子足臭jk番号 千仞雪被操漫画歪歪漫画 .
火影忍者纲手色 JapanPornMovies动漫 91爱妍小仙女在线播放 多国就俄罗斯局势表态 91麻豆精东乌鸦传媒 18禁爆乳网站 老年熟妇1 西藏2名干部被查 [NARUHO堂](全彩) 杭州老熟女私密处 .
三极片性生活 萌白酱自卫在线观看 亚洲精品国产aV成拍色拍麻豆
chineSe性Spy女厕 成人性爱一级黄片 亚洲男人和同 美人ママの勃起キープ枕営業 ~太いの一本挿れてくださいね~ 2 埃尔多安同意瑞典加入北约 各种姿势玩小处雌女 成人性生活三级片免费4466COm
. KK眼镜猥琐国模调教系列 日本波多野结黄色视频 不知火舞被❌自慰喷水爽
原神足控 丰田世纪SUV官方预告 欧美性爱49P
亚洲熟妇XXXXX色黄妇 体育生潮喷取精Chinese 深圳商场积水严重 VAM国漫3DMAX视频 .
黑人性犯罪糟蹋人妻 麻豆国产AV午床下闺蜜 最新女人另类2002
超变态玩弄折磨虐女系列视频 挠痒痒tk 电影消失演员表 婬乱的诊察室动漫01 西欧free性满足hd老熟妇 genshinsex无码网站
mdl夏晴子 . 金艺贞直播瑟妃 被男人吃奶很爽的毛叫床吧视频 口罩里面嘴被堵视频看故事 撒尿BBwBBwBBw喷水 神乳调教 多人吸奶电影 欧美AV➕无码➕高潮➕蜜臀
爆笑影片我不是故意的。 麻豆大陆无码AV
胖老太太女人裸体视频网站 . furry狼人榨精Gay网站 法院回应女子被法官抓伤面部 欧美18Gay 帅哥摔跤摸裆视频vk 本泽朋美国产一二三区 岛国动漫❌❌❌video 经典老汉GayOld2 国产老屄 日本娇小13XXXXHD 操哭furry网站 .
白丝自慰喷水白浆 生长板块和消亡板块岩石新老水状况 兔宝宝板材上的颜色是什么材质 OVA催○性指导5 女烈脚镣红色记忆 像新世界狂欢推荐
艾玛好色免费在线观看 性欧美图片 日本LESBIANXXXXHD 精灵 .
free 國模 警花被各种刑具调教高潮视频 德国黄色老胖女人免费看 变态性的暴行 永久成人黄色三级片 亚洲综合啪啪 巨大垂乳日本熟妇视频 索尼d6 随身听 肥臀熟女-OhPornTube
俄罗斯一油漆车间发生火灾 . 旧里番1~2姉恋在线观看百度云 性爱资源网 国产AV五十路六十路 马配种驴
性虎精品一区二 日本胖熊GayXXXX奥七部长 亲下面视频
女s女m女女调教视频系列 老熟女老女人AV 祢豆子爆乳乳液乱喷3d .
原神催眠调教黄高H文 十八岁第一次处破女 国产乱伦家庭网站 原神㊙️黄视频免费看 老头经典Gayoldman同性 日本❌❌❌Japanfree 越南xX❌╳妓女 男子家里做烟草零花钱20万?官方回应 观看老女人口交 八旬老头J八毛都白了
. 欧美绿奴喝尿cuckold视频 日本jjj 91视频 人妻×人妻
偷自福利拍 同人本子大全Onepiece 嫩小XXXXX性 成人抖音 91富二代 泰坦号观光艇5名乘员死亡
narutoPIXXXKonan丨vk . 一阴夹两阳是什么意思 白丝圣女呻吟浪荡堕落动漫 2kill4勒死资源 欧美18ⅩXOO极品 furry同性变态网站 欧美老妇人XXX高潮了 性少妇与黑人videoXXⅩ 京东传媒在线观看免费高清 好爽⋯好紧⋯再深一点男男小说 黄色板斗罗大陆
. 特斯拉回应召回百万辆车 色漫网站app免费 家庭乱伦网站视频 为母校打call什么意思
JulⅰeCaSh大白臀 Japanese成熟老妞 人妻系列无码老年人中出 帅老头T∨老头
欧美侵犯逼 3dAV网站 . 女尸vk 水滴偷拍换妻 黄片高晴无吗? 欧美阴阴道 大屁股女人的荫蒂 妓女爽死88888888 四川白天中年夫妇全程 熟女仑乱视频 美国丝袜脚交❌xX 91视频污污污 .
曝李玟遗体将被解剖 3D动漫jizz18 日本人japaniseXXHD j寡妇中国农村 国模冰 国产麻豆成人AV色影视在线
人虫尾交产虫H里番 cao porn福利社 王玉雯个人资料 按摩被中出在线 .
japaese老熟妇老熟女 日本漫画giantess XNXX日本免费观看 欧美丰满熟性XXXXXX 单亲乱XXXX 国产sm调教视频在线观看 麻豆水电工和少妇1080P 欧美BBw性色大片免费 日本小美女 黄色小说乱色
. 操逼,大片 日本影片r18网 调教SP菊眼浣肠 欧美黄色网站美女丝袜 老头Jj 美女祼体网站 中文字幕国产麻豆 南宁警方辟谣女子被当街强奸 莲花白发苦是啥原因 国产伦子XXXXX视频
.
I’m really enjoying the design and layout of
ykur blog. It’s a vwry eeasy oon the eyes whijch makes
it much ore enjoyable ffor mee tto cpme here and visit ore often. Did youu hiire out a dewigner
tto create your theme? Outstanding work!
Erica’s nude msn groupBrazil ass 2007 jelsoft enterprisess
ltdMy stepson iss gayMelodiie johnson nude picsBisexual gitl tank topSoftcokre milf lingetie picsOpen picture pussy wide.
View r kelly sex typeAmateuur wives iin pantyhoseEnglish shemale modelsWiffe swappping stgories and picturesEmmanule
chriquui nudePsytren henfai videoHairy mmen piucs and video.
Hot ssex with auntMadrd ggay hotelHot steamyy cockRickk bottoms
el cebtro caMstrkrft fist oof godd reviewsFrree
forced tto swwllow cum moviesShved pusasy vid teen. Asiian financial crisis world bankRecipe for asiawn chicken saladTesco een screen seattleHommo lingerie sexMature daiyBreaset cancer reurrence rates bby stageFree
gfs thumbs. Anal mussclesAdult entertainment ispand rhodeTesfudo
thhe terrapin andd virginBigg prfegnant breastsEbony lesbiasns sucking assesStormy daniels nude picsHow to builod a chubby box.
Malle homosexuall fictional storiesGay big dick tubeLeebanon prn moviesMature
sluts photosAdult legal site webAss hole trannyFemlale escorrs inn central pennsylvanis.
Mature male daddiesFree copck growthTroo sexyUnderfage
boys fucking young girlsAdulot entertainment florida in tampaAlessandro ambrosia nudeLaylaextreme pussy pump vid.
Hombres gaay pornoTotally spyyes pornJuicyy jaymes pornn starDrunk wonen nujde sexAdjlt survfivors
off mentwlly ill parentsWelding rule of thumb
costsNigerian adult. Gay couple germanyScooby dooo porn catoonsNorrth carrolina sex
paperYou8 bresxt bondageGirls realy have to peeBdsm epreuve soumisesSeelf
leevelling latex. Inuyasha hentai porn kagomeTraansgender modelsOlser lesebian tubeG
styring cumDoctoes examin naked girl videos metacafeRub him pantyhoseSucking your own dick techniches.
Guilty pleasure ppop songsLesbians grindinmg pussy oon assCowboy
hot cockBrazioian clip free sexFree teen puszy creampie10 secoond
orgasmWoken seex with dogs viss free. Listt asian capitalsBreast pump salesVintage beatles necklaceFinall
fantay 10 hentai games https://bit.ly/3GTXkNe Gabrieka bru
nudeShedevil sex.Pissed jeans peopke personThumb recognirion systemPenny flame teenFat gil mastrubating slujt loadBanng buss gayCum covered eyesXxxx
lino. Frree kim cattrall sex videosPorno boothSexual harassment ixaiah thomasMotrher
daughter nud naked photosFree user supported porn sitesLikee offf sexy underpants
womanFree xxx fackal pics. Young assian bdsm8th grrade sex
connecticutWoman seex beautyAmature mature housewifeEbony facials mpgsTeen sex young pornShakespeare bottom.
Woomen nnude underwearDiccipline pussy lickersMature classie photo galleryThirteen yeaar old grl sexyTeen onlinne ssex videosIs anal ssex
safe for menMomm oobs wityh son. Adult cat hhouse inn txForded swinger sexJapanese sports
sexFree mpeg women rapin meen seex moviesSpanish thyick pussyOlder mann
free poorn clipsFree gng bang vids sample.Fiind naked pic oof hhot cougarsTshirt whjoop assGerman nadyne model nakedCasting teen fuckSameera reddy sexy videosArchive
hustler magazineBerry frfee halle sex video. Brazilian addult model agencyChubby girl wettingAsioan irl inn white pantieBest faciaqls istFetiksh of
leotard tights 5Hot porn starr modelsPro’s of condoms. Zelea hentai comicDiary miulf mmrs zenFree
amature blowjob facialThee hair hustler salonLatex glpves vss petroleum productsFree homemade
interrracial videosEatinmg pussy slut.
Viiew sex videos tto fibd friends inn phoenixXxxx young giirls
suuck dickLesbo girls galleriesAmatuer babes nakedMaking hher wet fuckVintage clothijg
oon linePromethean board adult education. Tpu pornBiig beatiful women pornInvasive ductal carccinoma of breastTexzs mmens dicksMercedes ashley escortNirvana smells like a teen sporit lyricsOrganic faciwl skincare.
Cum on dagny showerSaginaw bay nudeTeen amateureAsshole tthe
card gameLesbuan mpeg strapAssk teenBoobs tthru a chair.
Dick pulsingBusty ai fuckRedhad mountsSeyspace latex packageMassage esa sexyVintage pstent leather purseNicee wet pussy pictures fingering.
Mother is a bdsm slave storiesBig boob bikinniVintage blowjob slutloadEroptic adult nursin relationshipBigg titts blonde pornAmateuyr cumshootsLeo lesbiaan turn on. Penthouyse
euro girls sexual historyTopless beach boobHazing
sexual maleParis rrocks pornNakjed kiss plumnp polishPreateen fuckBaby got boobs memphis monroe.
Pirates annd adult moviesArhhive cum facijal shotBojntiful booob curvesMaplee ridge stfip clubHentai
iphone videosSoft erotic porn fr girlosBlowmob sttripper videos.
Mature pussy pissingJessica drake bow jobScolre galleries breastTeen magazines badBeauty pageant cliips nudistMareie cavallier nudeSeex stim.
Exyy clitsDane bowers and jordan sexx tapeSwedishh artists / gayAspe skky nudeRockabilly
wwomen nudeSeex sinyles videoCherokee teacher ssex xvideos.
Open myy assholePajela andertson sexx scenes iin snapdragonPerfect
paiur boobsAmateur hardd homeStacion sexMilwaukee vintage clpthing storeFedom slave swap.
I’ve bedn expoloring ffor a bit for anny high-quality
articles oor weblog posts oon this kind of space .
Exploring in Yahoo I eventually stumbled upon this webb site.
Studyijng this information So i’m gglad to convey that I have an incrtedibly just right
uncanny feeling I fojnd out exactly what I needed. I such a llot without a ddoubt
will make suyre to do not fsil to remember tthis site and give itt a look on a relentless basis.
https://bit.ly/3te0Hb9
Good post! We aare linking to tbis partcularly great article onn ourr website.
Keeep upp the great writing.
Hurrah, that’s what I was exploring for, wht a material!
existingg here at thios weblog, thanks admun oof thhis site.
Hi there! Thhis iss my first visit too your blog!
We are a group off volumteers and starting a new initiastive in a
community inn tthe same niche. Your blog provided us useful
information to work on. You have don a extraordinar
job!
Magnificent bet ! I woul lke to apprsntice aat the same time ass you akend your web site,
how ccan i subscribe for a weblog website? The account aided mme a
applicable deal. I had been a little bit acquuainted of this our brroadcast provided shiny cledar idea
Freee hollywood sex tapesMinuude lesbioans girlsAveragee porn star wageFrree womens poen videosHot sexy nude tern girlsTranslation big tits att workFiist
full off boomstick review. Lesbians milkersGloc cockCoock and bupl the boathouseBeautiful fucking girlVirgin mobile compaatible
ringtonesVirgin mobile canada songLatex disguise.
Gatging vomit cumSandee west gatte anal tubeBangg free pic wife xxxHoww
does anorexia affects teen life2009 hustler calendersThumb rings and hasrley jewleryKatarina
witt nudes. Vivatar sexBig john stud gay thugResidential proggrams
for troubled adultsAdujlt halloween humor jokeMilwqaukee arult tamburitzansKiim kardashian free sex tapeHott naksd
preacher wife. Anal teachingFaust gayIttle virginsMcconaughey
ssex and the city1001 sxual positionsBilly bob thoornton penisPenis implant vido.
Anall schoogirlFree blonde pussyy picturesXxx porn vikew freeFree porn whhore onn whoreGretchen caroson upskiurt picsSex vixeo californiaBbw lesbien. Wommen wrestling men eroticMy bloody valentine nude girlAre miley cyrus boobs
realBarrier strip 33486Phhone sex lineArtemis nudeFreee gay porn sazmple videos.
Miss missouri teen internatiional ticketsSexx with medusaVintage toth powderAdult adltdisabilityreport ssa.govEmma wafson phitos niip slip upskirtLesbian brutal
sex videosPatt benetar ass nude. Dp sex for cashSeexy world wide fesmale
escortsGinny weaslley fuckHazel vintage dishwareHumor n sexx busty teen latinaVintag broadcast studio boardAre miicki james’s boobs real.
Cock ring exercisesXxx shemale adult hireSex for runnersDownlozd free
fucking clipsLovgely giirls nudeAdhlt pimple inside nos cureStrictly cum.
Nude scenss froom harold kumar guantanamoVenezula sex tour resortShort
sexy couples stories with picturesWhat iss great porn siteSex
snow videoCustom silk lingerieFree girdle feetish pics.
Csmetic facial surgery inn chicagoI fucked mmy soccer coachElisabeth
shuee naked inAnna kendrick fake nudes https://cutt.ly/bUogKC7 Nudee pic postingHaory wige pics.
Gayy male twinks and leafher dominenceBusty bikini babeWhy should i waait
for sexMaturre lesbos aand young lesbosBoys experiment witth ame sexBisexual
black maleShaved grannies. Gay monster pornFuck lookung wifeEros esxcorts san diegoMother nlaw blowjob videoParis hiltpn nuyde bathtubSwinging
lifesryle heidelberg minnesotaCarmen electra in lingerie.
Best amatteur threesomesVintage french lithographSex titfs
aass milfTumblr nnude natue bare gayMalle teen shavingPee wee
hermn photoPauley perrette pussy. Tight pussy get poundedSexy mims breast feedingBlond tkts washington ctFored ihto pantyhose storiesSexy teenagers in lingerieFreee llesbian milf and young videosRalph
laurenn fuck. Buick charlotte ddick gmmc keffer nnc pontiacSexyy hair silky shampooMedical fetish lare volume enemasNakd sster uptightShemale ucking black girlBlonde slim teen assYoou
tune videos mom fucks son. Hardcore naty teenHardcore older womanOld
yyoung lesbian thumbsHampster you tube amatuer vouyer sexAdul cerbral
palsy diapersBdsm cuut hair who womanFreee movies pussy. Breast
cancer and survivalUnsimulated blowjobAngel bblue
nudeJust teen site coldVintage naked womaan sictys picturesPussy movie thumbCirrhosis fluid brreasts penis.
Ethicl dilemas for teens leadershipFrree moom soon sexx taboo
vidVintagee orange ohler sinkMatire sex ladies for freeBeckham
david nakedd picNdubz sex free mp3 downloadPainful fiurst time anazl galleries.
My precioous virgins gretaUggly seex photosTall skinny naked ovber 40Ideal teen videosWomen flaashing tijts in carsFeenstr cherry 10 rule off thumbAnal gay massate
video. How tto use vibrator use picturesQuaad desert
aanal furyy privateAdulot dragon furryStyle advanced breast puump shoulderLijgerie
indianBlonde wwife tawkes oon 2 cocksPornstarr punichment.
Hardfcore gay assian videoAmateure teenns pornFree gay
muscle dickDog peeinhg on fire hydrasnt pictureFree sbian orgasm clipsEscot website uk cheapEstee
lauder amber nude bath and shower gel. Vintage arctic cat pantherWatch sex videeo without downloadFrree tiffany thisssen nudeLogin ended email username adult friencfinder personals compufer signedHow tto
orgasm feom oral sexHentai slime videoBiig mature areolas.
As pic prn str vval venisNatural vulvaRough sex home
mase videosDeep abal explorqtion toyAdult symptoms oof aspergersToorrent smokiing fetishSlow vibrator.
Vintage fiilm societySexyy naked ass sex amazingCalifornia gayy lompocPussey sexx machyine movieBreast implants
and tendernessIndikan maan and hotel fuckingLenndrum
breat centre. Rtpp gay escortSex in the cityy 2 reviewTwwink wowIving deep throatAdult sex flash ganes swimWiffe videos adultMesican gorl deepthroat.
Faty vaginaAdultt bokstore south bendStopping teen violence iin schooolFrree
porn movfie off pasris hiltonKim kardashian naked photosFree ude college camSucck ooff eliot.
Adult flash games2Uk adult payy pper view sitesFial fantasu pornHoot brazilian lesbiansTrick with
thumb tipNobody nudeNaked women howing off vaginas. Bbbw dress tgpCaloifornia quarter midxget racingFrree biig
boobs adult mopvie clipsAnetta ass keyFree xxx wet panties porn picsTyler clementus ssex tapeYpung pussies.
What a mateerial of un-ambiguity and preserveness of precious experience regarding unexpefted emotions.
hi!,I really luke yolur writinbg vry so much! proportion we keep inn touch
more aboout you article onn AOL? I require an expert on this house too nravel my problem.
May bee tgat iis you! Having a look forward too peer you.
I’mtruly enjoying thee design and lasyout of your website.
It’sa very easy on thee eyess which makes it muc mor pleasant foor mme tto come here
aand visit mopre often. Did yoou hire out a develoer to
create your theme? Outstanding work!
https://bit.ly/2Tsc2rM
I don’t even know how I ended upp rigjt here, holwever I assumed this
post waas oncee great. I do nnot know wwho you aree buut definitely you
aree going to a famous bloggerr if you aren’t already.
Cheers!
Great blog rijght here! Addiionally your websiote loads up verey fast!
Whhat web host are youu thhe usage of? Can I am gtting your affiliate
linmk to yojr host? I want my web site loaded up aas fast as ours lol
Hey There. I foumd your weblog tthe use off msn. This iis ann extremely smargly wrotten article.
I wilpl be sure to bookmark itt and come bawck to read more of yoyr
useful information. Thanks for the post. I’ll certainly comeback.
tadalafil 5mg pills tadalafil 40mg generic ed pills that work
isotretinoin 10mg canada isotretinoin 20mg brand purchase zithromax online
order azipro 500mg buy omnacortil no prescription order gabapentin 600mg generic
lasix 40mg cheap doxycycline 100mg us ventolin 4mg canada
buy generic vardenafil buy zanaflex pills for sale purchase hydroxychloroquine for sale
ramipril 10mg uk buy etoricoxib online arcoxia online
order levitra 20mg without prescription order vardenafil 10mg online cheap order plaquenil 200mg pill
where to buy asacol without a prescription buy astelin generic purchase avapro sale
order benicar 20mg generic buy depakote buy cheap depakote
how to buy temovate generic temovate amiodarone order
buy carvedilol 25mg generic brand cenforce 100mg buy aralen without a prescription
diamox uk buy diamox 250mg online oral azathioprine 25mg
digoxin for sale how to buy digoxin molnupiravir 200 mg cheap
buy generic naprosyn 250mg naprosyn canada buy prevacid 15mg online
purchase proventil pill protonix 40mg for sale purchase pyridium online
cheap montelukast 10mg buy generic singulair order generic avlosulfon 100mg
baricitinib usa buy baricitinib 2mg without prescription lipitor online order
order adalat 30mg without prescription buy allegra 180mg sale fexofenadine 180mg pills
where to buy amlodipine without a prescription buy prilosec generic buy omeprazole generic
buy dapoxetine misoprostol buy online xenical 120mg tablet
buy lopressor generic buy methylprednisolone 4 mg can i buy methylprednisolone online
order diltiazem 180mg order zovirax generic zyloprim online
how to buy triamcinolone order generic clarinex 5mg buy loratadine online cheap
rosuvastatin 20mg generic crestor 10mg cost buy generic motilium
ampicillin 250mg brand buy metronidazole generic metronidazole drug
order sumycin 250mg sale cyclobenzaprine order where to buy lioresal without a prescription
buy sulfamethoxazole pills generic cleocin 300mg order cleocin 150mg sale
toradol buy online buy toradol generic buy inderal online
buy plavix no prescription order warfarin generic warfarin online
order erythromycin fildena sale buy tamoxifen 20mg online cheap
metoclopramide 10mg sale cost losartan 25mg buy nexium online
cost rhinocort ceftin uk careprost drug
buy topamax imitrex 25mg usa order generic levofloxacin
order robaxin 500mg without prescription methocarbamol oral purchase suhagra generic
buy dutasteride medication buy avodart 0.5mg online cheap cost mobic
sildenafil 100mg generic sildenafil 50mg us buy cheap generic estrace
generic celecoxib 200mg celebrex pills zofran 8mg ca
buy lamotrigine generic buy lamotrigine paypal prazosin 1mg cheap
spironolactone 100mg sale aldactone 100mg ca valacyclovir online order
propecia 5mg drug sildenafil 100mg price order generic viagra 50mg
tretinoin gel brand purchase tadalafil sale order avana 200mg generic
cialis coupon walmart cheap cialis 20mg buy sildenafil 50mg
oral tadalafil 20mg order indomethacin 75mg pills oral indocin
cialis mail order usa diflucan cost non prescription erection pills
lamisil generic cefixime uk buy amoxicillin pills for sale
sulfasalazine 500mg uk benicar 10mg without prescription verapamil 120mg without prescription
order arimidex order biaxin 500mg sale buy clonidine sale
depakote over the counter buy generic imdur over the counter order imdur
order meclizine online cheap order antivert 25 mg generic buy generic minomycin
buy imuran 25mg online cheap order digoxin 250 mg generic cheap telmisartan
buy molnunat 200mg sale naproxen 500mg price omnicef 300mg usa
buy erectile dysfunction meds sildenafil mail order viagra 50mg cost
buy prevacid 30mg for sale lansoprazole 15mg us protonix canada
buy ed pills online order cialis 5mg pill cialis 5mg
fexofenadine 120mg uk brand fexofenadine amaryl 4mg uk
terazosin 1mg without prescription buy hytrin 5mg pills cialis generic name
etoricoxib 60mg without prescription mesalamine brand buy astelin sprayer
CEX.IO accepts both debit cards and credit cards and you can use them either for instant fiat deposits and direct crypto purchases. To buy Bitcoin with a debit card instantly, open the Buy Sell page, choose the amount and click Buy. On the next step, you can add a new payment card to your CEX.IO account or pay for crypto with the one you’ve linked before. Want to buy crypto? Simply send funds to your self-custodial wallet with wire, ACH, or credit card. Give instructions directly to your bank. Here’s what they need to know: As a new DeFi user, are you wondering how to transfer the funds in your Ethereum wallet to your bank or crypto exchange? This guide will walk you through this process. Truestory Valentine’s Day exclusive limited-time offers are starting to go live. It’s a good opportunity to gift experiences or gift cards to your loved ones, from nice dinners, spa days, or even a solve-a-mystery experience to do together.
http://fit-girl.co.kr/bbs/board.php?bo_table=free&wr_id=38503
If you’re interested in forking Ethereum, you can follow Alchemy’s step-by-step guide on how to fork Ethereum mainnet with a free Alchemy account, HardHat, and a command line interface (CLI). There are two types of forks: We’ll use Ganache CLI to fork the Mainnet; Ganache CLI uses ethereumjs to simulate full client behavior making development on Ethereum faster, easier, and safer. In layman’s terms, a cryptocurrency exchange is a place where you meet and exchange cryptocurrencies with another person. The exchange platform (i.e. Binance) acts as a middleman – it connects you (your offer or request) with that other person (the seller or the buyer). With a brokerage, however, there is no “other person” – you come and exchange your crypto coins or fiat money with the platform in question, without the interference of any third party. When considering cryptocurrency exchange rankings, though, both of these types of businesses (exchanges and brokerages) are usually just thrown under the umbrella term – exchange. This is done for the sake of simplicity.
order avapro 150mg without prescription brand buspirone 10mg buspar 5mg generic
amiodarone 100mg price order generic phenytoin 100 mg order phenytoin sale
Please let me know if you’re looking for a writer for your weblog. You have some really good articles and I believe I would be a good asset. If you ever want to take some of the load off, I’d really like to write some material for your blog in exchange for a link back to mine. Please send me an email if interested. Many thanks!
oxytrol for sale online elavil 10mg sale alendronate 70mg over the counter
biltricide online buy praziquantel 600mg generic periactin generic
order macrodantin 100mg sale motrin 600mg without prescription nortriptyline cost
buy fluvoxamine 50mg generic purchase duloxetine for sale buy duloxetine 20mg generic
buy glucotrol generic where to buy nootropil without a prescription betamethasone medication
I liked up to you’ll receive carried out right here. The cartoon is attractive, your authored subject matter stylish. nonetheless, you command get bought an nervousness over that you would like be turning in the following. ill definitely come more in the past once more as precisely the same nearly very steadily inside of case you shield this increase.
Thank you for sharing with us, I believe this website really stands out : D.
acetaminophen 500mg drug order paxil 10mg generic buy pepcid 20mg
buy clomipramine 25mg online cheap progesterone 100mg pill prometrium 100mg generic
tacrolimus price order ropinirole 1mg sale buy ropinirole 1mg generic
tinidazole order online order bystolic 5mg for sale order nebivolol online
diovan order online where to buy ipratropium without a prescription buy generic combivent
purchase calcitriol pill order tricor 160mg online order tricor 200mg sale
Perfectly composed subject material, thankyou for entropy.
buy decadron 0,5 mg pill zyvox where to buy order starlix 120mg pills
oxcarbazepine without prescription order ursodiol 300mg online urso for sale online
order capoten 25mg online buy tegretol without prescription order carbamazepine 200mg generic
order bupropion 150mg without prescription bupropion 150 mg sale order strattera
buy ciprofloxacin online cheap purchase cefadroxil for sale cefadroxil generic
seroquel 100mg cost how to buy zoloft lexapro 20mg canada
where can i buy combivir buy lamivudine without prescription order accupril without prescription
zoosk dating: https://datingtopreview.com/# – tinder login
cost propecia tablets propecia without rx
Read information now.
cost of propecia without dr prescription get propecia no prescription
Read now.
https://propecia1st.science/# cheap propecia without rx
cost mobic without rx order generic mobic online can i order cheap mobic pill
https://propecia1st.science/# cost of propecia tablets
fluoxetine over the counter buy letrozole no prescription letrozole generic
where can i buy cheap mobic for sale: buy cheap mobic online – buying generic mobic price
amoxicillin medicine where to buy amoxicillin 500mg – amoxicillin price without insurance
cost of generic propecia without insurance cost propecia tablets
get generic mobic pill: order generic mobic tablets – can i buy cheap mobic online
northwest pharmacy canada canada drugs
frumil drug purchase clindamycin online purchase zovirax generic
Read information now.
pills for ed: what is the best ed pill – new treatments for ed
Everything what you want to know about pills.
erection pills: ed pills – ed drug prices
https://propecia1st.science/# get generic propecia without insurance
order generic mobic prices: can i buy mobic price – how to buy cheap mobic prices
can i order generic mobic without dr prescription mobic pills can i get mobic pills
https://propecia1st.science/# get generic propecia without a prescription
amoxicillin 500 mg tablet price: amoxicillin 500mg no prescription amoxicillin canada price
buy amoxicillin online cheap amoxicillin generic – amoxicillin 775 mg
can i order cheap mobic pills: can i buy generic mobic price – cheap mobic tablets
reliable canadian online pharmacy canadian pharmacy online reviews
where to buy mobic without prescription: cost cheap mobic without prescription – where buy cheap mobic no prescription
where can you get amoxicillin: how to get amoxicillin amoxicillin 775 mg
Actual trends of drug.
cost cheap propecia without dr prescription buy propecia no prescription
All trends of medicament.
amoxicillin 1000 mg capsule: http://amoxicillins.com/# how to get amoxicillin
buy bisoprolol generic buy ethambutol 1000mg pills terramycin 250mg sale
buy generic propecia no prescription home
generic propecia pills get propecia
https://pharmacyreview.best/# escrow pharmacy canada
amoxicillin medicine over the counter: amoxicillin no prescription amoxicillin without prescription
canada pharmacy online: canadian pharmacy no scripts – canadian pharmacy 365
valaciclovir 500mg tablet buy famvir no prescription order ofloxacin
https://mexpharmacy.sbs/# mexican pharmaceuticals online
http://certifiedcanadapharm.store/# canadian pharmacy 1 internet online drugstore
canadapharmacyonline legit: safe canadian pharmacies – canadian pharmacy ed medications
mexican online pharmacies prescription drugs: mexican rx online – mexican rx online
https://mexpharmacy.sbs/# best online pharmacies in mexico
canadian mail order pharmacy: buy canadian drugs – best online canadian pharmacy
cefpodoxime 100mg drug purchase flixotide generic flixotide online
canadian pharmacy antibiotics: my canadian pharmacy – northern pharmacy canada
http://indiamedicine.world/# top 10 online pharmacy in india
https://indiamedicine.world/# top online pharmacy india
levetiracetam uk buy keppra 1000mg for sale cost of viagra 100mg
mexican pharmaceuticals online: reputable mexican pharmacies online – purple pharmacy mexico price list
india pharmacy: Online medicine home delivery – top online pharmacy india
http://certifiedcanadapharm.store/# real canadian pharmacy
https://indiamedicine.world/# reputable indian pharmacies
vipps approved canadian online pharmacy: canadian online pharmacy – canadian pharmacy online store
world pharmacy india: cheapest online pharmacy india – indian pharmacy online
http://certifiedcanadapharm.store/# northwest pharmacy canada
canada ed drugs: canada pharmacy – is canadian pharmacy legit
http://mexpharmacy.sbs/# mexican rx online
buying prescription drugs in mexico: mexico drug stores pharmacies – medication from mexico pharmacy
http://certifiedcanadapharm.store/# canadian world pharmacy
canadadrugpharmacy com: best rated canadian pharmacy – canadian pharmacies online
order zaditor 1mg generic doxepin price cheap tofranil
https://mexpharmacy.sbs/# mexico pharmacies prescription drugs
http://stromectolonline.pro/# ivermectin australia
https://gabapentin.pro/# medicine neurontin 300 mg
zithromax 500 mg lowest price online zithromax cost zithromax 500 without prescription
http://azithromycin.men/# how to get zithromax
ivermectin 18mg: ivermectin lotion cost – ivermectin 5 mg
Thank you for the good writeup. It in fact was a amusement account it.
Look complicated to far introduced agreeable from you!
However, how could we keep up a correspondence?
First of all I want to say awesome blog! I had a quick question which I’d like to ask if you do not mind.
I was interested to know how you center yourself and clear your thoughts prior to writing.
I’ve had a hard time clearing my mind in getting my ideas out.
I truly do enjoy writing however it just seems like the first 10 to 15 minutes tend to be lost simply just trying to figure out how to begin. Any suggestions or
tips? Thanks!
hello!,I really like your writing so a lot! share we keep
up a correspondence extra about your post on AOL? I need an expert in this space to unravel
my problem. Maybe that’s you! Taking a look forward to see
you.
Very soon this site will be famous among all blog visitors, due to it’s
good articles
http://gabapentin.pro/# neurontin buy online
buy neurontin canada: neurontin generic brand – buy brand neurontin
http://azithromycin.men/# can i buy zithromax over the counter
order zithromax over the counter buy zithromax 1000 mg online where can i buy zithromax medicine
buy acarbose without prescription prandin cost order fulvicin 250mg online
ivermectin 3 mg tabs: ivermectin stromectol – oral ivermectin cost
http://stromectolonline.pro/# ivermectin buy
https://azithromycin.men/# zithromax price south africa
aspirin 75 mg sale where to buy zovirax without a prescription zovirax online order
https://ed-pills.men/# buy erection pills
buy antibiotics from india: buy antibiotics from india – get antibiotics quickly
https://antibiotic.guru/# buy antibiotics online
best ed treatment: new ed pills – ed drugs list
http://ciprofloxacin.ink/# buy ciprofloxacin tablets
Great post. I used to be checking constantly this blog and I am impressed!
Extremely useful information specially the remaining
part 🙂 I take care of such info much. I used to
be seeking this particular information for a long time.
Thanks and best of luck.
http://lisinopril.pro/# lisinopril cost us
Thanks for sharing excellent informations. Your website is very cool. I am impressed by the details that you have on this site. It reveals how nicely you understand this subject. Bookmarked this web page, will come back for extra articles. You, my friend, ROCK! I found simply the information I already searched all over the place and simply could not come across. What an ideal web site.
https://misoprostol.guru/# cytotec pills buy online
http://avodart.pro/# avodart for sale
meloset drug buy danazol 100mg online buy danazol without a prescription
http://ciprofloxacin.ink/# cipro
order dipyridamole 100mg pills gemfibrozil medication where can i buy pravastatin
https://ciprofloxacin.ink/# ciprofloxacin generic
https://misoprostol.guru/# cytotec pills buy online
https://ciprofloxacin.ink/# buy cipro cheap
I’m really enjoying the design and layout of your blog.
It’s a very easy on the eyes which makes it much more enjoyable for me to come here and visit more often. Did you hire out a developer to create your theme?
Fantastic work!
https://lipitor.pro/# lipitor purchase
dydrogesterone 10 mg drug buy dapagliflozin 10mg online cheap order generic jardiance
reputable mexican pharmacies online mexico pharmacies prescription drugs medication from mexico pharmacy
reputable indian online pharmacy: best online pharmacy india – india online pharmacy
http://mexicanpharmacy.guru/# mexico drug stores pharmacies
medication canadian pharmacy: legitimate canadian online pharmacies – canadian pharmacies that deliver to the us
florinef 100 mcg brand buy imodium 2mg generic loperamide 2mg canada
buy monograph 600mg generic colospa 135mg buy pletal 100mg pills
Hi there mates, how is everything, and what you wish for to say regarding this article,
in my view its actually remarkable in support of me.
It’s going to be end of mine day, however before finish I am reading this enormous post to improve my knowledge.
To announce true to life scoop, follow these tips:
Look for credible sources: https://w1000w.com/wp-content/pages/where-da-at-doe-news-anchor.html. It’s important to guard that the newscast roots you are reading is reliable and unbiased. Some examples of reliable sources subsume BBC, Reuters, and The Modish York Times. Interpret multiple sources to pick up a well-rounded understanding of a particular statement event. This can support you return a more complete facsimile and keep bias. Be hep of the perspective the article is coming from, as constant respected hearsay sources can have bias. Fact-check the low-down with another source if a news article seems too unequalled or unbelievable. Forever be persuaded you are reading a known article, as tidings can substitute quickly.
Nearby following these tips, you can fit a more in the know dispatch reader and more intelligent apprehend the world here you.
certainly like your web site however you need to take a look at the spelling on several of
your posts. Many of them are rife with spelling
problems and I in finding it very troublesome to inform the reality on the other
hand I’ll surely come again again.
What you said was actually very logical. However, think about this, what if you typed
a catchier title? I mean, I don’t want to tell you how to run your blog, however what if you added a headline that grabbed people’s attention? I mean Neon Flying Squid – Joyseafood.ltd is a little vanilla.
You might glance at Yahoo’s front page and watch how they write article titles to get people to
open the links. You might add a related video or a picture or two to grab people interested about everything’ve got to say.
In my opinion, it would bring your blog a little livelier.
prasugrel us tolterodine 1mg pills tolterodine 2mg cheap
I have beeen browsing online more thann 3 hours today, yet I never found
anny intgeresting article liike yours. It’s pretty worth ennough for me.
In myy view, iff aall sitte owners and bllggers mzde good content as yyou did, thhe webb will bbe a lott more useful than eve before.
mestinon 60 mg oral brand rizatriptan 5mg maxalt 10mg without prescription
Hi there! I simply want to give you a big thumbs up for the great info you’ve got right here on this post.
I will be returning to your website for more soon.
bet365 Casino No Deposit Bonus This website is using a security service to protect itself from online attacks. The action you just performed triggered the security solution. There are several actions that could trigger this block including submitting a certain word or phrase, a SQL command or malformed data. These no deposit voucher codes are a word or a string of random letters and numbers that act as a gatekeeper to the promotion – simply enter this code when prompted and you can enjoy free bets, free spins, money back offers and so much more besides. This is a great promo because you’ll have $1,000 in free bets to play with, helping you add to your balance in your online casino account. It’s a better offer than a no-deposit bonus because it’s more lucrative, putting you in a better position to convert these funds into more winnings.
http://www.chungdamfeel.com/bbs/board.php?bo_table=free&wr_id=124348
Of course! The majority of online poker sites have either mobile apps or mobile-friendly versions of their websites so you can play online poker wherever you are. All you’ll need is an internet connection to access the same poker games available to desktop and mac users, on the go! Third party monitoring of online poker sites is also done by eCOGRA (e Commerce Online Gaming Regulation and Assurance), which is a grassroots organization formed by poker players, bettors and gamblers to monitor online gaming and ensure poker sites are fair. Regular independent testing is done and on its website and its audits are published. Any signs of rigging would show up here and a non-compliant site would be red-flagged as a rigged poker site. source Not all poker sites are safe. Before you share your personal details with online casinos, always conduct your due diligence to ensure that you land on a safe poker platform. All our picks have been tested and verified as reliable online poker sites.
What’s up to every body, it’s my first pay a quick visit of this weblog; this website consists
of amazing and truly excellent material in support of visitors.
Thanks for your personal marvelous posting! I actually enjoyed reading it, you
are a great author. I will be sure to bookmark your blog and will
eventually come back someday. I want to encourage you to definitely continue your great work, have a
nice day!
cost vasotec 5mg order duphalac bottles buy lactulose generic
xalatan without prescription latanoprost online buy buy exelon no prescription
reputable indian pharmacies: international pharmacy india – world pharmacy india
Altogether! Conclusion info portals in the UK can be unendurable, but there are many resources available to cure you mark the unexcelled identical for the sake of you. As I mentioned formerly, conducting an online search representing https://www.wellpleased.co.uk/wp-content/pages/what-happened-to-sam-brock-nbc-news-the-latest.html “UK scuttlebutt websites” or “British story portals” is a pronounced starting point. Not but purposefulness this grant you a encompassing slate of report websites, but it choice also lend you with a heartier pact of the current news scene in the UK.
In the good old days you obtain a liber veritatis of embryonic news portals, it’s critical to gauge each one to determine which overwhelm suits your preferences. As an benchmark, BBC Intelligence is known in place of its intention reporting of intelligence stories, while The Trustee is known quest of its in-depth analysis of political and popular issues. The Self-governing is known championing its investigative journalism, while The Times is known for its vocation and wealth coverage. By way of entente these differences, you can select the information portal that caters to your interests and provides you with the hearsay you want to read.
Additionally, it’s worth all in all local expos‚ portals with a view explicit regions within the UK. These portals lay down coverage of events and dirt stories that are relevant to the область, which can be firstly accommodating if you’re looking to keep up with events in your close by community. For occurrence, shire news portals in London include the Evening Standard and the Londonist, while Manchester Evening Hearsay and Liverpool Echo are stylish in the North West.
Overall, there are many tidings portals available in the UK, and it’s important to do your digging to find the joined that suits your needs. By evaluating the unconventional low-down portals based on their coverage, dash, and editorial perspective, you can select the song that provides you with the most related and engrossing news stories. Meet luck with your search, and I anticipate this information helps you discover the correct dope portal suitable you!
canadian pharmacy india: Online medication home delivery – mail order pharmacy india
top 10 pharmacies in india: Online medication home delivery – world pharmacy india
There is definately a great deal to find out about this subject.
I like all of the points you made.
purchase betahistine generic buy haldol 10 mg generic probalan without prescription
buy generic premarin 0.625mg buy dostinex 0.25mg online cheap viagra 50mg tablet
purchase omeprazole buy prilosec for sale buy metoprolol pills
buy cialis 5mg online cheap viagra tablets buy generic sildenafil 100mg
where to buy telmisartan without a prescription order molnunat 200mg generic purchase molnunat for sale
cenforce generic order cenforce sale order generic chloroquine 250mg
top 10 online pharmacy in india: indianpharmacy com – indianpharmacy com
When some one searches for his essential thing, therefore he/she
needs to be available that in detail, thus that thing is maintained over here.
I’ve been exploring for a bit for any high quality articles or
weblog posts in this sort of space . Exploring in Yahoo I ultimately
stumbled upon this web site. Reading this info So i’m happy to express that I’ve an incredibly excellent uncanny feeling I
discovered exactly what I needed. I most surely will make
certain to don?t forget this website and give it a look on a
continuing basis.
top 10 online pharmacy in india: best online pharmacy india – world pharmacy india
modafinil us buy modafinil 100mg sale order prednisone online
You could definitely see your skills in the work you write.
The world hopes for even more passionate writers like you who are not
afraid to mention how they believe. Always follow your heart.
Hurrah, that’s what I was looking for, what a information! existing here at this blog, thanks admin of this web
site.
international pharmacy online canadian international pharmacy canadian mail order prescriptions
Thanks for sharing such a good thinking, post is nice, thats why i have read it fully
buy omnicef 300 mg pill cefdinir 300mg drug lansoprazole online
wonderful submit, very informative. I ponder why the opposite experts of this sector don’t notice
this. You should continue your writing. I’m sure, you have a
huge readers’ base already!
Thank you for any other informative site. The place else could I
am getting that type of info written in such a perfect way?
I’ve a undertaking that I am simply now running on, and I have been at the glance out for such information.
oral isotretinoin amoxicillin 250mg for sale order azithromycin 500mg
Paragraph writing is also a fun, if you know after that you can write otherwise it is complex to write.
Thank you for any other informative site. Where else could I get that type of information written in such a perfect way?
I have a project that I’m just now running on, and I’ve been at the look out for such information.
purchase azithromycin online cost omnacortil order gabapentin 600mg pills
Heya exceptional blog! Does running a blog like this require a lot of work?
I have absolutely no understanding of computer programming however
I had been hoping to start my own blog soon. Anyways, if you have any
recommendations or tips for new blog owners please share.
I know this is off topic nevertheless I just needed to ask.
Thanks a lot!
buy cheap lipitor albuterol 100 mcg without prescription order amlodipine for sale
http://farmaciabarata.pro/# farmacias online seguras en espaГ±a
versandapotheke versandkostenfrei versandapotheke versandkostenfrei versandapotheke deutschland
http://farmaciaonline.men/# farmacia online senza ricetta
online roulette game real money play online roulette for fun order furosemide 40mg generic
https://esfarmacia.men/# farmacias online seguras
cheap pantoprazole 40mg order protonix 20mg pill phenazopyridine 200 mg drug
online-apotheken: versandapotheke – online-apotheken
I like looking through a post that will make men and women think.
Also, thank you for allowing for me to comment!
https://edpharmacie.pro/# Pharmacies en ligne certifiГ©es
Thanks a lot for sharing this with all folks you
actually recognize what you are talking approximately! Bookmarked.
Kindly additionally consult with my site =). We could have a
hyperlink alternate arrangement between us
best online poker sites online gambling slots games ventolin inhalator without prescription
I am extremely impressed together with your writing talents and also with the layout on your blog.
Is that this a paid topic or did you customize it your self?
Anyway stay up the nice high quality writing, it’s uncommon to see
a nice weblog like this one today..
Pretty great post. I simply stumbled upon your blog and wished to say that I have really enjoyed browsing your
weblog posts. In any case I will be subscribing to your rss feed and I’m hoping
you write again soon!
acheter medicament a l etranger sans ordonnance: Pharmacie en ligne sans ordonnance
Hey there! Do you use Twitter? I’d like to follow you if
that would be okay. I’m undoubtedly enjoying your blog and look
forward to new posts.
best online casino free slots online ivermectina
buy symmetrel 100mg generic where to buy atenolol without a prescription order dapsone generic
mexican pharmaceuticals online: mexican online pharmacies prescription drugs – medication from mexico pharmacy
canadian pharmacy meds: canada pharmacy online – canadian drug stores
playing poker online for money order synthroid 75mcg online cheap buy synthroid medication
Their international patient care is impeccable. mexican pharmaceuticals online: buying prescription drugs in mexico online – mexican online pharmacies prescription drugs
mexico drug stores pharmacies: purple pharmacy mexico price list – medicine in mexico pharmacies
Hi, Neat post. There is an issue along with your web site in internet
explorer, may check this? IE nonetheless is the
marketplace chief and a huge component of other people
will leave out your great writing because of this problem.
whoah this blog is magnificent i like reading your articles.
Stay up the good work! You recognize, lots of individuals are hunting around for this
information, you can aid them greatly.
Always a pleasant experience at this pharmacy. top 10 online pharmacy in india: reputable indian pharmacies – indian pharmacy paypal
reputable mexican pharmacies online: mexican pharmaceuticals online – mexican mail order pharmacies
indianpharmacy com: cheapest online pharmacy india – indian pharmacies safe
clomid over the counter order imuran online cheap buy imuran 50mg pills
Top-notch medications sourced globally. world pharmacy india: best india pharmacy – buy medicines online in india
purchase medrol buy aristocort pills for sale where to buy triamcinolone without a prescription
buying from online mexican pharmacy: buying from online mexican pharmacy – mexico drug stores pharmacies
buy prescription drugs from canada cheap: canadian pharmacy meds – canadian drugs online
It’s an awesome paragraph in favor of all
the web people; they will get benefit from it I am sure.
Their online prescription system is so efficient. reputable indian online pharmacy: best online pharmacy india – pharmacy website india
purchase levitra pill where can i buy zanaflex buy tizanidine medication
canada pharmacy world: cheapest pharmacy canada – canadian pharmacy 24
The pharmacists always take the time to answer my questions. top 10 pharmacies in india: online pharmacy india – indian pharmacy
buy drugs from canada: cheap canadian pharmacy – ed meds online canada
I am really loving the theme/design of your weblog.
Do you ever run into any internet browser compatibility problems?
A couple of my blog readers have complained about my website not working
correctly in Explorer but looks great in Safari.
Do you have any ideas to help fix this problem?
buy prescription drugs from india: indian pharmacy – Online medicine home delivery
A pharmacy that truly values its patrons. purple pharmacy mexico price list: buying prescription drugs in mexico – mexican drugstore online
phenytoin 100 mg price order ditropan 2.5mg pill purchase oxytrol online
aceon 4mg pills buy coversum pills for sale order fexofenadine 180mg
Pretty great post. I just stumbled upon your blog and wished to say that I’ve really loved browsing
your blog posts. After all I’ll be subscribing to your
feed and I hope you write again very soon!
Consistently excellent, year after year. http://doxycyclineotc.store/# doxycycline prescription cost uk
They always keep my medication history well-organized. http://edpillsotc.store/# ed pills online
best erection pills cheap ed drugs the best ed pills
Get information now. http://azithromycinotc.store/# where can i buy zithromax medicine
Their digital prescription service is innovative and efficient. https://azithromycinotc.store/# zithromax buy online no prescription
A true champion for patients around the world. zithromax online paypal: buy zithromax – zithromax 250 mg australia
buy doxycycline 11554 buy doxycycline doxycycline online paypal
http://edpillsotc.store/# new ed pills
Я всегда мечтал о том, чтобы попробовать свои силы в онлайн казино, и вот мой шанс наконец пришел благодаря сайту caso-slots.com. С его помощью я нашел все популярные казино и даже список тех, где предлагают бонусы на первый депозит. Пришло время веселья!
order baclofen generic elavil 10mg ca order generic toradol
Hi there to all, how is all, I think every one is getting more from this
web page, and your views are nice designed
for new visitors.
A person essentially assist to make severely articles I might
state. That is the very first time I frequented your web page and
to this point? I surprised with the analysis you made to create this particular publish extraordinary.
Wonderful job!
A stalwart in international pharmacy services. http://drugsotc.pro/# affordable pharmacy
buy claritin no prescription buy dapoxetine 90mg online buy priligy 30mg sale
Leading the charge in international pharmacy standards. http://drugsotc.pro/# online pharmacy australia free delivery
mexican pharmacy buy drugs at mexican pharmacy mexican mail order pharmacies
Παρασκευή 10 Μαΐου, 2019, 09:36 Δεν είναι μυστικό ότι τα διαδικτυακά καζίνο ανέκαθεν εμπλέκονταν στη μουσική βιομηχανία. Τώρα αποτελούν ένα μεγάλο μέρος της! Η σχέση μεταξύ των διαδικτυακών καζίνο και της μουσικής είναι τόσο ισχυρή που θα δείτε ακόμα πολλές συνεργασίες να επισυνάπτονται στο μέλλον. Ακολουθούν μερικά μόνο παραδείγματα για το πώς αυτή η σχέση έχει πάρει άλλες διαστάσεις: Το Πρόσωπο με το σχήμα του όρθιου τριγώνου
http://topdogs1.co.kr/bbs/board.php?bo_table=free&wr_id=271542
online casino bonus χωρις καταθεση Ειδικευόμαστε στις ηλεκτρολογικές εργασίες και εγκαταστάσεις από το 1980. Με στόχο το άψογο αποτέλεσμα στην σωστή τιμή και πάντα την καλύτερη εξυπηρέτηση αποκτήσαμε την εμπιστοσύνη των πελατών μας. Κατά τη διάρκεια της έρευνας, στην οποία ήταν παρόντες οι δύο ιδιοκτήτες του υποστατικού, εντοπίστηκαν 6 πύργοι ηλεκτρονικών υπολογιστών και ένας φορητός ηλεκτρονικός υπολογιστής, τα οποία παραλήφθηκαν για περαιτέρω επιστημονικές εξετάσεις, καθώς και χρηματικό ποσό.
Their international health campaigns are revolutionary. https://indianpharmacy.life/# п»їlegitimate online pharmacies india
order ozobax pills oral ketorolac toradol pills
I trust them with all my medication needs. https://indianpharmacy.life/# reputable indian pharmacies
They simplify the complexities of international prescriptions. https://drugsotc.pro/# best canadian pharmacy
online pharmacy without scripts canadian mail order pharmacy the peoples pharmacy
What’s up mates, how is everything, and what you desire to say about this post, in my
view its genuinely remarkable designed for
me.
alendronate cheap macrodantin order online macrodantin 100 mg us
I appreciate their late hours for those unexpected needs. http://drugsotc.pro/# canadian pharmacy price checker
buy glimepiride for sale generic cytotec 200mcg order etoricoxib pills
Woah! I’m really digging the template/theme of this website.
It’s simple, yet effective. A lot of times it’s very hard to get that “perfect balance” between superb
usability and visual appeal. I must say that you’ve done a great
job with this. Additionally, the blog loads very fast for me on Internet explorer.
Excellent Blog!
Global expertise that’s palpable with every service. http://canadapharmacy.cheap/# reliable canadian pharmacy reviews
pharmacy no prescription: mexican pharmacy without prescription – canadian drugstore pharmacy
inderal 20mg drug buy inderal 20mg pill plavix where to buy
Hi there! This is kind of off topic but I need some
help from an established blog. Is it very difficult to set up your
own blog? I’m not very techincal but I can figure things
out pretty fast. I’m thinking about creating my own but I’m not
sure where to begin. Do you have any ideas or suggestions?
Many thanks
mexican online pharmacies prescription drugs and mexico online pharmacy – reputable mexican pharmacies online
reputable mexican pharmacies online : mexico online pharmacy – purple pharmacy mexico price list
nortriptyline for sale online buy nortriptyline 25mg sale buy panadol 500mg
best online pharmacies in mexico – pharmacy in mexico – buying prescription drugs in mexico
buying prescription drugs in mexico : buying prescription drugs in mexico online – pharmacies in mexico that ship to usa
http://stromectol24.pro/# minocycline capsules 100mg
order generic warfarin metoclopramide 10mg pill buy metoclopramide 10mg sale
canadian pharmacy oxycodone: canadian pharmacy pro – canada ed drugs
https://stromectol24.pro/# generic ivermectin for humans
buy orlistat pill asacol 400mg pills order diltiazem 180mg for sale
indian pharmacy: indian pharmacy paypal – indian pharmacy
https://canadapharmacy24.pro/# my canadian pharmacy reviews
I think the admin of this site is actually working hard in favor of
his web page, since here every information is quality based data.
top online pharmacy india: indian pharmacies safe – canadian pharmacy india
http://stromectol24.pro/# ivermectin stromectol
best online pharmacy india: indian pharmacy paypal – best online pharmacy india
where to buy famotidine without a prescription buy prograf pills prograf for sale
https://stromectol.icu/# how much is ivermectin
http://plavix.guru/# Plavix 75 mg price
order astelin 10ml sale acyclovir us avapro pills
Plavix generic price: plavix best price – cheap plavix antiplatelet drug
valtrex script online: buy valtrex online – valtrex tablets online
http://valtrex.auction/# how to get valtrex prescription
esomeprazole 20mg uk order mirtazapine 30mg pill topamax 100mg pills
Hurrah, that’s what I was searching for, what a material!
present here at this web site, thanks admin of this web site.
paxlovid india: paxlovid pharmacy – paxlovid india
https://stromectol.icu/# order stromectol online
buy kamagra online usa: Kamagra tablets – Kamagra 100mg price
Levitra 10 mg best price Buy Vardenafil 20mg Levitra 20 mg for sale
https://kamagra.icu/# Kamagra tablets
http://kamagra.icu/# Kamagra 100mg price
Buy Levitra 20mg online Vardenafil buy online Cheap Levitra online
http://cialis.foundation/# Cialis 20mg price
imitrex 25mg oral avodart pills buy dutasteride online cheap
http://viagra.eus/# viagra without prescription
oral allopurinol 100mg zyloprim 100mg brand buy crestor 20mg pill
buy Viagra online Viagra Tablet price generic sildenafil
https://viagra.eus/# Generic Viagra for sale
buy zantac 300mg pill order mobic order celecoxib pill
https://kamagra.icu/# Kamagra 100mg
Kamagra Oral Jelly buy kamagra online usa Kamagra Oral Jelly
buspin over the counter buy buspirone 10mg online cheap cordarone 200mg cost
http://levitra.eus/# п»їLevitra price
https://kamagra.icu/# Kamagra 100mg price
cheap kamagra sildenafil oral jelly 100mg kamagra cheap kamagra
https://kamagra.icu/# Kamagra 100mg price
http://levitra.eus/# Vardenafil online prescription
order generic tamsulosin 0.4mg simvastatin price zocor over the counter
sildenafil 50 mg price generic sildenafil buy Viagra online
http://levitra.eus/# Buy Vardenafil 20mg online
purchase motilium generic generic coreg 6.25mg cheap tetracycline 500mg
https://kamagra.icu/# Kamagra Oral Jelly
Kamagra Oral Jelly super kamagra buy Kamagra
http://levitra.eus/# Cheap Levitra online
cheap spironolactone 25mg spironolactone 25mg drug propecia 1mg oral
mexican mail order pharmacies: mexico pharmacies prescription drugs – mexican pharmacy mexicanpharmacy.company
canadian pharmacy: canadian pharmacy service – legitimate canadian online pharmacies canadapharmacy.guru
http://canadapharmacy.guru/# canadapharmacyonline legit canadapharmacy.guru
medication from mexico pharmacy: buying prescription drugs in mexico – purple pharmacy mexico price list mexicanpharmacy.company
thesis writing services essay helper academic writing uk
canadian family pharmacy: canadian drugs – pharmacy in canada canadapharmacy.guru
reputable mexican pharmacies online: buying prescription drugs in mexico online – п»їbest mexican online pharmacies mexicanpharmacy.company
https://mexicanpharmacy.company/# mexican mail order pharmacies mexicanpharmacy.company
http://canadapharmacy.guru/# canadianpharmacymeds canadapharmacy.guru
I quite like reading an article that will make people think.
Also, many thanks for permitting me to comment!
mexico pharmacies prescription drugs: mexican online pharmacies prescription drugs – mexican pharmaceuticals online mexicanpharmacy.company
Your style is very unique compared to other people I’ve read stuff from.
Many thanks for posting when you have the opportunity, Guess I
will just book mark this page.
buying prescription drugs in mexico: buying from online mexican pharmacy – mexico drug stores pharmacies mexicanpharmacy.company
http://mexicanpharmacy.company/# best online pharmacies in mexico mexicanpharmacy.company
purchase forcan without prescription buy ampicillin no prescription buy cipro 500mg generic
buy medicines online in india: top online pharmacy india – online pharmacy india indiapharmacy.pro
http://canadapharmacy.guru/# canadian pharmacy canadapharmacy.guru
buy sildenafil 50mg generic aurogra order online estrace 1mg canada
pharmacy in canada: canadian family pharmacy – real canadian pharmacy canadapharmacy.guru
recommended canadian pharmacies: canadian pharmacy 24 – onlinecanadianpharmacy 24 canadapharmacy.guru
online canadian drugstore: canadian online pharmacy – canadian pharmacy scam canadapharmacy.guru
https://canadapharmacy.guru/# canadian online pharmacy canadapharmacy.guru
metronidazole 200mg tablet cheap cephalexin buy cephalexin without prescription
best online pharmacy india: top online pharmacy india – india online pharmacy indiapharmacy.pro
https://canadapharmacy.guru/# onlinecanadianpharmacy canadapharmacy.guru
http://canadapharmacy.guru/# northwest pharmacy canada canadapharmacy.guru
canadian pharmacy ratings: buying from canadian pharmacies – online canadian pharmacy review canadapharmacy.guru
http://indiapharmacy.pro/# cheapest online pharmacy india indiapharmacy.pro
pharmacies in mexico that ship to usa: mexico drug stores pharmacies – buying prescription drugs in mexico mexicanpharmacy.company
top online pharmacy india: indian pharmacy – top 10 pharmacies in india indiapharmacy.pro
buy lamictal 200mg online purchase nemazole online buy mebendazole pills
http://mexicanpharmacy.company/# buying prescription drugs in mexico mexicanpharmacy.company
https://canadapharmacy.guru/# legit canadian pharmacy canadapharmacy.guru
reputable indian online pharmacy: best online pharmacy india – reputable indian online pharmacy indiapharmacy.pro
clindamycin usa purchase fildena pills cheap fildena 100mg
canadian pharmacies online: canadian pharmacy ed medications – my canadian pharmacy canadapharmacy.guru
http://indiapharmacy.pro/# online pharmacy india indiapharmacy.pro
Online medicine order: world pharmacy india – top online pharmacy india indiapharmacy.pro
http://mexicanpharmacy.company/# pharmacies in mexico that ship to usa mexicanpharmacy.company
http://mexicanpharmacy.company/# buying from online mexican pharmacy mexicanpharmacy.company
purple pharmacy mexico price list: buying from online mexican pharmacy – mexican border pharmacies shipping to usa mexicanpharmacy.company
order tretinoin gel online order avana 100mg generic purchase stendra pills
http://indiapharmacy.pro/# indian pharmacy online indiapharmacy.pro
get cheap clomid without insurance: order generic clomid no prescription – cheap clomid pills
http://prednisone.digital/# buy prednisone 10mg online
buy tamoxifen 20mg generic tamoxifen pills symbicort without prescription
prednisone 20mg prescription cost: prednisone brand name india – 50mg prednisone tablet
https://amoxil.world/# buy amoxicillin online without prescription
cheap doxycycline online: doxycycline medication – 100mg doxycycline
https://amoxil.world/# amoxicillin 500 mg brand name
http://propecia.sbs/# order propecia
generic doxycycline: order doxycycline online – buy doxycycline online 270 tabs
buy amoxicillin from canada: amoxicillin price canada – generic for amoxicillin
We’re a group of volunteers and opening a new scheme in our community.
Your website provided us with valuable information to work on. You’ve done an impressive job and our entire
community will be grateful to you.
can you get generic clomid online: where can i get clomid pill – get clomid without a prescription
https://prednisone.digital/# generic over the counter prednisone
tadalafil 10mg generic tadacip 20mg us indocin 75mg without prescription
cost of cheap propecia without insurance: buying propecia without prescription – order propecia pills
generic axetil order axetil for sale generic methocarbamol 500mg
http://clomid.sbs/# where can i buy clomid pills
prednisone 40mg: buy prednisone nz – where to buy prednisone 20mg
http://amoxil.world/# buy amoxicillin
buy doxycycline online uk: how to buy doxycycline online – buy doxycycline cheap
http://doxycycline.sbs/# where to purchase doxycycline
cheap propecia price: cost of cheap propecia tablets – buy cheap propecia pill
http://doxycycline.sbs/# doxycycline medication
order generic propecia without insurance: order propecia without prescription – cheap propecia price
http://prednisone.digital/# prednisone online india
https://withoutprescription.guru/# buy cheap prescription drugs online
mexico drug stores pharmacies: medicine in mexico pharmacies – mexican drugstore online
lamisil online terbinafine for sale online online gambling casinos
real viagra without a doctor prescription usa: generic viagra without a doctor prescription – prescription drugs online
http://canadapharm.top/# recommended canadian pharmacies
mexico pharmacies prescription drugs: purple pharmacy mexico price list – mexican border pharmacies shipping to usa
https://mexicopharm.shop/# mexican drugstore online
ed pills that really work: erectile dysfunction drugs – generic ed pills
https://canadapharm.top/# northwest pharmacy canada
my favorite writer essay essay writing on football suprax 100mg tablet
legal to buy prescription drugs from canada: generic viagra without a doctor prescription – non prescription erection pills
essays done for you write my thesis blackjack games
http://mexicopharm.shop/# mexican border pharmacies shipping to usa
canadian pharmacy ltd: Legitimate Canada Drugs – online canadian pharmacy reviews
https://withoutprescription.guru/# sildenafil without a doctor’s prescription
order cheap propecia no prescription: generic propecia price – cheap propecia without insurance
best ed pills at gnc: ed pills otc – herbal ed treatment
purchase amoxicillin generic brand arimidex biaxin 250mg canada
best ed medication: new ed drugs – cheap erectile dysfunction pills online
viagra without doctor prescription: buy prescription drugs from india – non prescription ed drugs
buy propecia no prescription: generic propecia without dr prescription – buy cheap propecia prices
cost rocaltrol 0.25mg labetalol 100 mg pill order tricor 200mg online
https://withoutprescription.guru/# prescription drugs online without
Levitra 20 mg for sale: Levitra 10 mg best price – Buy Vardenafil 20mg online
https://tadalafil.trade/# tadalafil 20mg pills
sildenafil 100 mg best price sildenafil 100 coupon sildenafil 100 mg tablet
http://levitra.icu/# Buy Levitra 20mg online
tadalafil 20mg price in india: tadalafil canadian pharmacy price – buy tadalafil uk
http://kamagra.team/# cheap kamagra
Levitra 20 mg for sale buy Levitra over the counter Buy Vardenafil 20mg
ed treatment drugs: erection pills viagra online – best pills for ed
prescription medication for adult acne cause of pimples in adults trileptal 600mg cost
buy catapres 0.1mg online cheap meclizine 25mg pills buy tiotropium bromide 9mcg sale
sildenafil tablets online in india: can i buy sildenafil over the counter in canada – where can i buy sildenafil 100mg
http://kamagra.team/# п»їkamagra
buy Kamagra Kamagra 100mg Kamagra 100mg
https://edpills.monster/# natural ed medications
https://levitra.icu/# Buy Levitra 20mg online
tadalafil capsules 21 mg: tadalafil for sale from india – cheap 10 mg tadalafil
Cheap Levitra online Vardenafil online prescription п»їLevitra price
erectile dysfunction drug: ed pills cheap – erection pills that work
tadalafil 40 mg online india: tadalafil cheapest price – how much is tadalafil
http://tadalafil.trade/# tadalafil tablets price in india
alfuzosin sale best allergy pill for itching common heartburn medication prescription
http://doxycycline.forum/# doxy 200
buy cipro online Ciprofloxacin online prescription cipro 500mg best prices
order minocycline order ropinirole 2mg buy ropinirole cheap
where to buy amoxicillin 500mg: amoxicillin generic – purchase amoxicillin 500 mg
amoxicillin online purchase: buy amoxil – amoxicillin tablet 500mg
http://lisinopril.auction/# lisinopril 20mg pill
order amoxicillin online no prescription purchase amoxicillin online amoxicillin buy no prescription
can you buy doxycycline over the counter in nz: doxycycline buy online – doxycycline tetracycline
Hello i am kavin, its my first time to commenting anyplace, when i read this piece of writing i thought i could also create
comment due to this good article.
http://ciprofloxacin.men/# ciprofloxacin 500mg buy online
ciprofloxacin 500mg buy online Buy ciprofloxacin 500 mg online buy cipro online canada
zithromax without prescription: zithromax z-pak – where to get zithromax
cipro pharmacy: Get cheapest Ciprofloxacin online – buy cipro online without prescription
buy sleep aids online fda hair net requirements 2024 prescription diet pills online pharmacy
https://amoxicillin.best/# amoxicillin cost australia
zestril price in india Buy Lisinopril 20 mg online lisinopril prescription cost
cheap letrozole 2.5 mg order generic aripiprazole aripiprazole 20mg drug
zestril 10 mg price in india: cheapest price for lisinopril india – lisinopril for sale online
Taking responsibility for one’s actions contributes to a mature and
respectful relationship.
http://amoxicillin.best/# 875 mg amoxicillin cost
buy amoxil: buy amoxil – cost of amoxicillin
prescription for amoxicillin: purchase amoxicillin online – amoxicillin from canada
buy ciprofloxacin over the counter buy ciprofloxacin over the counter buy cipro online canada
withdrawals from smoking cigarettes fda approval smoking cessation medication list of painkillers by name
https://doxycycline.forum/# where to buy doxycycline online
You can certainly see your expertise within the work you write.
The world hopes for more passionate writers like you who aren’t afraid to say how they believe.
Always go after your heart.
buy cipro online without prescription: ciprofloxacin – buy cipro
http://indiapharmacy.site/# india pharmacy
п»їbest mexican online pharmacies: best online pharmacy – mexican online pharmacies prescription drugs
purchase medroxyprogesterone online microzide uk purchase microzide without prescription
pharmacy world: cheap drugs online – overseas no rx drugs online
meds canadian compounding pharmacy cheapest online pharmacy fda approved canadian online pharmacies
mumbai-wife-in-5-star-hotel-with-lover
milf-eating-creamy-pussy
hot-poso
ureshidrea
http://canadiandrugs.store/# best canadian online pharmacy
Online medicine order: top online pharmacy india – indian pharmacy paypal
canadian pharmacy online no prescription: Top mail order pharmacies – top rated online pharmacies
vitamins to prevent herpes outbreaks inhaler costs list current pharmaceutical commercials
http://canadiandrugs.store/# www canadianonlinepharmacy
top canadian online pharmacy: buy prescription drugs online – best canadian drugstore
reputable indian pharmacies mail order pharmacy india top 10 pharmacies in india
best online pharmacies in mexico: mexican pharmacy – mexican mail order pharmacies
periactin 4mg uk buy cyproheptadine 4 mg oral nizoral 200mg
http://clomid.club/# where to buy generic clomid online
buy ventolin without prescription: ventolin 200 mg – buy ventolin over the counter nz
name of antifungal drugs cheapest online prescription refill best high blood pressure meds
https://claritin.icu/# ventolin mexico
buy wellbutrin online: buy wellbutrin – wellbutrin prescription australia
https://paxlovid.club/# buy paxlovid online
order duloxetine 20mg purchase provigil online cheap provigil 100mg
how to get neurontin: cheap gabapentin – purchase neurontin canada
I’m not sure exactly why but this weblog is loading incredibly slow for me.
Is anyone else having this issue or is it a problem on my end?
I’ll check back later on and see if the problem still exists.
http://gabapentin.life/# neurontin 300mg tablet cost
how long do stomach ulcers last duodenal ulcer vs stomach ulcer common bacteria found in uti
150 wellbutrin tablets: Buy Wellbutrin XL online – wellbutrin 150 xl
http://wellbutrin.rest/# can i purchase wellbutrin without prescription in canada
wellbutrin 75 mg tablet: Wellbutrin prescription – wellbutrin 100mg tablets
https://claritin.icu/# buy cheap ventolin online
order generic phenergan buy erectile dysfunction meds ivermectin 3mg over counter
Good day! This post could not be written any better!
Reading this post reminds me of my good old room mate! He
always kept chatting about this. I will forward this post to him.
Fairly certain he will have a good read. Thanks
for sharing!
Paxlovid over the counter: paxlovid club – п»їpaxlovid
http://clomid.club/# clomid without insurance
morning after pill buy online how to stop ejacilating early best sperm increase pills
Howdy, I do believe your site may be having
browser compatibility issues. Whenever I look at your site in Safari, it looks fine however when opening
in Internet Explorer, it’s got some overlapping issues.
I simply wanted to provide you with a quick heads up!
Apart from that, wonderful website!
farmacie online affidabili: avanafil generico – farmacia online più conveniente
comprare farmaci online con ricetta avanafil prezzo in farmacia farmacie online sicure
farmacia online miglior prezzo: Dove acquistare Cialis online sicuro – farmacie online affidabili
farmacia online miglior prezzo: Farmacie che vendono Cialis senza ricetta – farmacia online migliore
https://kamagrait.club/# comprare farmaci online con ricetta
viagra generico recensioni: viagra consegna in 24 ore pagamento alla consegna – viagra online in 2 giorni
acquisto farmaci con ricetta: Farmacie che vendono Cialis senza ricetta – farmacia online migliore
viagra subito viagra generico viagra originale in 24 ore contrassegno
farmacia online migliore: farmacia online spedizione gratuita – farmaci senza ricetta elenco
miglior sito dove acquistare viagra: viagra senza ricetta – viagra originale in 24 ore contrassegno
deltasone 5mg ca deltasone 10mg for sale buy amoxil 1000mg online
esiste il viagra generico in farmacia: viagra consegna in 24 ore pagamento alla consegna – viagra subito
https://farmaciait.pro/# migliori farmacie online 2023
top farmacia online: dove acquistare cialis online sicuro – farmacie on line spedizione gratuita
viagra consegna in 24 ore pagamento alla consegna: viagra senza ricetta – viagra prezzo farmacia 2023
migliori farmacie online 2023 Farmacie a milano che vendono cialis senza ricetta acquistare farmaci senza ricetta
farmacie online autorizzate elenco: avanafil generico prezzo – farmacia online miglior prezzo
stomach acid reducer medicine treatment for dry mucous membranes does gas medicine stop farting
comprare farmaci online con ricetta: kamagra gel – migliori farmacie online 2023
farmacia online: kamagra gold – migliori farmacie online 2023
viagra pfizer 25mg prezzo: viagra online spedizione gratuita – alternativa al viagra senza ricetta in farmacia
farmaci senza ricetta elenco: cialis prezzo – acquistare farmaci senza ricetta
https://sildenafilit.bid/# viagra generico in farmacia costo
farmacie online sicure: kamagra oral jelly – farmacia online migliore
viagra generico in farmacia costo alternativa al viagra senza ricetta in farmacia dove acquistare viagra in modo sicuro
farmacia online miglior prezzo: Dove acquistare Cialis online sicuro – comprare farmaci online all’estero
farmaci senza ricetta elenco: farmacia online migliore – top farmacia online
farmaci senza ricetta elenco: farmacia online migliore – farmacia online
farmacia online migliore: farmacia online miglior prezzo – migliori farmacie online 2023
http://farmaciait.pro/# comprare farmaci online con ricetta
acquistare farmaci senza ricetta: farmacia online – farmacie online affidabili
farmacia online cialis generico consegna 48 ore farmacia online
comprare farmaci online all’estero: avanafil generico prezzo – acquistare farmaci senza ricetta
Keep up the fantastic work, I read few articles on this internet site and I believe that your web site is real interesting and holds lots of fantastic info .
farmacia online più conveniente: Avanafil farmaco – comprare farmaci online con ricetta
Thanks for sharing your thoughts. I truly appreciate your efforts and I will be waiting for your further post thank you once again.
viagra consegna in 24 ore pagamento alla consegna: alternativa al viagra senza ricetta in farmacia – cialis farmacia senza ricetta
farmacia online senza ricetta: avanafil prezzo in farmacia – farmacia online senza ricetta
farmacia online senza ricetta kamagra gel farmacie on line spedizione gratuita
https://tadalafilit.store/# farmaci senza ricetta elenco
viagra acquisto in contrassegno in italia: sildenafil prezzo – alternativa al viagra senza ricetta in farmacia
farmacia online: Tadalafil generico – farmacia online più conveniente
purchase urso ursodiol 300mg price buy zyrtec 5mg for sale
I was suggested this blog by my cousin. I’m not sure whether
this post is written by him as no one else know such detailed about my difficulty.
You are incredible! Thanks!
acquisto farmaci con ricetta: Dove acquistare Cialis online sicuro – comprare farmaci online con ricetta
farmaci senza ricetta elenco: farmacia online miglior prezzo – farmacie online sicure
Hello, this weekend is pleasant in favor of me, because this occasion i
am reading this impressive educational paragraph here at my home.
YSLDA Vegas Slot Gam… Being a beauty editor, I have gone the retinol route, tried and abandoned many a light peeling lotion, exfoliating, glycolic and everything else. “Hack” can mean a lot of things in the modern context. Be it the breach of confidential information, or the simple life hacks that make your life better. Grains and Gains Challenge: turn the slots into harvesting machines!Crank up your yields with as many winning spins as possible and get loads of: Twists Boosters Free Spins Bonus: bit.ly 45RCfPt (Safe mode: bonus can be claimed in Browser) bit.ly 3QE3fxQ (Mobile) Many of you guys already know about Lucky patcher app. Lucky Patcher is an all in one rooted app for Android that can do many tasks if you have root access. Do you wish to block ads on your Android? Do you want to remove unnecessary stock apps from your smartphone? or do you want to bypass license verification on paid Android apps? Here we have an app that can do this for you!! Let’s see what exactly it is.
https://jeffreyrbmv987463.dailyblogzz.com/23372235/virgin-mobile-online-casino
Semua mesin judi slot88 disini sangat fairplay tanpa ada modus tipuan, karena sampai sejauh ini hanya Juaraslot88 yang menggunakan sistem RNG dimana putaran mesin slot berjalan secara otomatis sehingga tak akan ada settingan sekecil apapun. Tingkat kekalahan dalam permainan slot88 sudah pasti jauh lebih sedikit, tak heran bila member setia Juaraslot88 selalu berhasil mendapatkan kemenangan dan jackpot hingga jutaan bahkan ratusan juta rupiah setiap harinya. Daya pikat Jackpot slot terbesar sudah menanti anda, hiburan sederhana sangat menarik sekali untuk ditaruhkan para pecinta slot di Indonesia. Hal ini menjadi peluang terbaik buat kalian semua untuk bisa bermain judi slot gacor terbesar ini, menyediakan kemudahan dalam bermain dengan system pastinya sangat terpercaya. Kalian bisa mendapatkan penghasilan tambahan hanya dengan bersantai – santai sambil menikmati game judi slot88 mudah menang, tidak perlu mengeluarkan uang untuk pergi ke casino, kalian juga bisa menyiapkan segalanya sebelum kalian bermain siapkan posisi yang nyaman dan juga beberapa cemilan untuk menemani kalian bermain game.
farmacia online piГ№ conveniente farmacia online miglior prezzo comprare farmaci online con ricetta
farmacia online: kamagra – farmaci senza ricetta elenco
http://kamagraes.site/# farmacia online
viagra online cerca de la coruГ±a: comprar viagra – comprar viagra sin gastos de envГo
http://kamagraes.site/# farmacia barata
farmacias baratas online envГo gratis Levitra sin receta farmacia online internacional
https://kamagraes.site/# farmacia online internacional
https://kamagraes.site/# farmacias online seguras en espaГ±a
https://tadalafilo.pro/# farmacia online envÃo gratis
farmacia barata: Levitra sin receta – farmacias online baratas
https://tadalafilo.pro/# farmacia online 24 horas
My coder is trying to persuade me to move to .net
from PHP. I have always disliked the idea because
of the costs. But he’s tryiong none the less. I’ve been using Movable-type on several websites
for about a year and am nervous about switching to another
platform. I have heard very good things about blogengine.net.
Is there a way I can import all my wordpress posts into it?
Any kind of help would be really appreciated!
http://kamagraes.site/# farmacia online envÃo gratis
farmacias online baratas Levitra precio farmacia online barata
There’s definately a lot to find out about this topic. I love
all of the points you’ve made.
http://vardenafilo.icu/# farmacia online envÃo gratis
https://farmacia.best/# farmacia online madrid
https://kamagraes.site/# farmacia online
farmacia online envГo gratis: farmacia envio gratis – farmacias online baratas
http://farmacia.best/# farmacia online internacional
buy strattera generic buy cheap generic strattera order generic sertraline 50mg
farmacia online barata comprar cialis online sin receta farmacia online madrid
https://kamagraes.site/# farmacia online envГo gratis
http://tadalafilo.pro/# farmacias online seguras en españa
http://sildenafilo.store/# sildenafilo 100mg precio farmacia
Thank you, I’ve just been searching for information about this topic for a while and yours is the greatest I’ve discovered till now. But, what in regards to the conclusion? Are you sure concerning the supply?
http://farmacia.best/# farmacia online 24 horas
https://farmacia.best/# farmacias baratas online envÃo gratis
farmacias online seguras en espaГ±a vardenafilo farmacias online seguras
https://vardenafilo.icu/# п»їfarmacia online
http://sildenafilo.store/# viagra online cerca de zaragoza
farmacia 24h: farmacia envio gratis – п»їfarmacia online
http://tadalafilo.pro/# farmacias online baratas
http://sildenafilo.store/# viagra para hombre venta libre
https://sildenafilo.store/# sildenafilo cinfa sin receta
https://tadalafilo.pro/# farmacia online barata
http://tadalafilo.pro/# farmacias baratas online envГo gratis
sildenafilo precio farmacia comprar viagra contrareembolso 48 horas comprar viagra en espaГ±a
http://vardenafilo.icu/# farmacias online seguras
farmacia 24h: cialis en Espana sin receta contrareembolso – farmacia 24h
http://kamagraes.site/# farmacia 24h
order lexapro generic where can i buy fluoxetine revia 50mg ca
buy cheap generic lasix order ventolin 2mg without prescription cheap albuterol
https://tadalafilo.pro/# farmacia barata
https://sildenafilo.store/# viagra online cerca de bilbao
http://vardenafilo.icu/# farmacias online baratas
farmacias online baratas: kamagra gel – farmacia online madrid
http://tadalafilo.pro/# farmacias online baratas
http://tadalafilo.pro/# farmacias online seguras en españa
sildenafilo cinfa sin receta sildenafilo precio viagra entrega inmediata
https://tadalafilo.pro/# farmacia online internacional
http://tadalafilo.pro/# farmacia barata
farmacias online baratas: se puede comprar kamagra en farmacias – farmacia online barata
https://kamagraes.site/# farmacias online seguras
https://farmacia.best/# farmacia envГos internacionales
http://farmacia.best/# farmacias online seguras en españa
farmacia envГos internacionales Cialis sin receta п»їfarmacia online
https://farmacia.best/# farmacia online barata
http://farmacia.best/# farmacia online envÃo gratis
farmacias online seguras: farmacia online envio gratis – farmacia barata
https://sildenafilo.store/# comprar viagra online en andorra
oral amoxiclav purchase clomid generic clomid over the counter
http://farmacia.best/# farmacias online baratas
oral combivent dexona pills buy zyvox generic
farmacias online seguras en espaГ±a Cialis precio п»їfarmacia online
https://vardenafilo.icu/# farmacia barata
http://vardenafilo.icu/# farmacia envÃos internacionales
http://farmacia.best/# farmacia online internacional
https://kamagrafr.icu/# acheter médicaments à l’étranger
farmacias online baratas: comprar cialis online seguro opiniones – farmacias online seguras
https://cialissansordonnance.pro/# Pharmacie en ligne sans ordonnance
Pharmacie en ligne France kamagra oral jelly acheter medicament a l etranger sans ordonnance
https://viagrasansordonnance.store/# Viagra homme sans prescription
Viagra homme sans ordonnance belgique: Meilleur Viagra sans ordonnance 24h – Viagra vente libre pays
https://cialissansordonnance.pro/# Pharmacie en ligne livraison gratuite
https://levitrafr.life/# Pharmacie en ligne livraison 24h
viagra para hombre venta libre: sildenafilo precio – sildenafilo sandoz 100 mg precio
https://levitrafr.life/# acheter medicament a l etranger sans ordonnance
п»їViagra sans ordonnance 24h Meilleur Viagra sans ordonnance 24h Viagra homme sans ordonnance belgique
https://pharmacieenligne.guru/# Pharmacie en ligne pas cher
Pharmacie en ligne pas cher: pharmacie en ligne – pharmacie ouverte
order nateglinide 120mg sale atacand 16mg over the counter candesartan 16mg brand
farmacia online madrid: kamagra 100mg – farmacias online baratas
http://kamagrafr.icu/# acheter medicament a l etranger sans ordonnance
http://cialissansordonnance.pro/# pharmacie en ligne
Sildenafil teva 100 mg sans ordonnance Viagra gГ©nГ©rique sans ordonnance en pharmacie Viagra homme sans ordonnance belgique
https://pharmacieenligne.guru/# Pharmacie en ligne livraison 24h
order starlix sale buy atacand 8mg without prescription atacand 8mg uk
п»їpharmacie en ligne: Medicaments en ligne livres en 24h – Acheter mГ©dicaments sans ordonnance sur internet
http://pharmacieenligne.guru/# pharmacie ouverte 24/24
farmacia online 24 horas: farmacias online seguras – farmacia online barata
https://levitrafr.life/# Pharmacie en ligne livraison gratuite
https://levitrafr.life/# pharmacie en ligne
http://levitrafr.life/# pharmacie en ligne
acheter mГ©dicaments Г l’Г©tranger Pharmacie en ligne France Pharmacie en ligne pas cher
Pharmacies en ligne certifiГ©es: cialis generique – acheter mГ©dicaments Г l’Г©tranger
farmacia 24h: farmacias online seguras – farmacia online madrid
http://pharmacieenligne.guru/# Acheter médicaments sans ordonnance sur internet
buy generic levitra online plaquenil brand buy plaquenil cheap
https://levitrafr.life/# Pharmacies en ligne certifiées
internet apotheke online apotheke versandkostenfrei online apotheke deutschland
tegretol 200mg pills lincomycin 500 mg for sale how to buy lincomycin
http://cialiskaufen.pro/# online apotheke deutschland
internet apotheke cialis preise versandapotheke
https://potenzmittel.men/# п»їonline apotheke
http://apotheke.company/# online apotheke versandkostenfrei
Appreciate the recommendation. Will try it out.
versandapotheke online apotheke gГјnstig online apotheke preisvergleich
My brother suggested I would possibly like this website.
He was totally right. This post truly made my day.
You can not imagine just how so much time I had spent for this information! Thank you!
Hello, i believe that i noticed you visited my website so i got here to return the
choose?.I’m trying to to find things to improve my site!I suppose its good enough to use some of your concepts!!
Definitely believe that which you said. Your favorite justification appeared to be on the net the easiest
thing to be aware of. I say to you, I definitely get irked while people consider
worries that they plainly do not know about. You
managed to hit the nail upon the top as well as defined out the whole thing without having side-effects , people
could take a signal. Will probably be back to
get more. Thanks
The study has to include the evaluation of the estrogen and progesterone receptor and HER2 gene expression I, A cialis price
Wow, wonderful blog layout! How long have you been blogging for?
you make blogging look easy. The overall look of your website is magnificent, let
alone the content!
Hi there! Do you know if they make any plugins to assist with Search Engine Optimization? I’m trying to get my blog to rank for some targeted keywords but I’m not seeing very good gains.
If you know of any please share. Appreciate it!
Thank you, I have just been looking for information approximately this subject for ages and
yours is the best I have discovered till now. However, what in regards to the bottom line?
Are you certain about the supply?
http://kamagrakaufen.top/# internet apotheke
cenforce 100mg for sale buy glucophage 500mg for sale order generic metformin 500mg
An interesting discussion is worth comment. I think that you should write more on this topic, it might not be a taboo subject but generally people are not enough to speak on such topics. To the next. Cheers
http://apotheke.company/# versandapotheke versandkostenfrei
Viagra kaufen ohne Rezept legal viagra generika Viagra online bestellen Schweiz
versandapotheke deutschland: cialis rezeptfreie kaufen – versandapotheke versandkostenfrei
http://potenzmittel.men/# versandapotheke deutschland
Sildenafil kaufen online viagra kaufen ohne rezept legal In welchen europäischen Ländern ist Viagra frei verkäuflich
https://viagrakaufen.store/# Viagra rezeptfreie Schweiz bestellen
order duricef online cost cefadroxil combivir us
best mexican online pharmacies medicine in mexico pharmacies mexican border pharmacies shipping to usa
mexico drug stores pharmacies pharmacies in mexico that ship to usa best online pharmacies in mexico
atorvastatin over the counter norvasc usa purchase lisinopril without prescription
mexico drug stores pharmacies medicine in mexico pharmacies mexico drug stores pharmacies
https://mexicanpharmacy.cheap/# buying prescription drugs in mexico
mexican border pharmacies shipping to usa mexico drug stores pharmacies mexican pharmacy
mexican online pharmacies prescription drugs best online pharmacies in mexico buying prescription drugs in mexico
http://mexicanpharmacy.cheap/# best online pharmacies in mexico
pharmacies in mexico that ship to usa mexican border pharmacies shipping to usa mexican border pharmacies shipping to usa
buying prescription drugs in mexico online mexican pharmaceuticals online medication from mexico pharmacy
mexican pharmaceuticals online mexican mail order pharmacies buying prescription drugs in mexico
reputable mexican pharmacies online mexican pharmaceuticals online mexican pharmaceuticals online
best online pharmacies in mexico mexican mail order pharmacies reputable mexican pharmacies online
https://mexicanpharmacy.cheap/# buying prescription drugs in mexico online
http://mexicanpharmacy.cheap/# buying prescription drugs in mexico online
buying prescription drugs in mexico online buying prescription drugs in mexico online best online pharmacies in mexico
http://mexicanpharmacy.cheap/# mexican pharmaceuticals online
mexican pharmacy mexico pharmacies prescription drugs buying prescription drugs in mexico
mexican pharmaceuticals online mexican drugstore online buying prescription drugs in mexico online
https://mexicanpharmacy.cheap/# reputable mexican pharmacies online
mexico pharmacies prescription drugs best mexican online pharmacies mexican pharmacy
medication from mexico pharmacy mexican rx online buying from online mexican pharmacy
purple pharmacy mexico price list purple pharmacy mexico price list medicine in mexico pharmacies
medicine in mexico pharmacies mexican drugstore online mexican pharmacy
http://mexicanpharmacy.cheap/# medicine in mexico pharmacies
buying prescription drugs in mexico medicine in mexico pharmacies mexican mail order pharmacies
Very nice article. I certainly appreciate this
website. Keep writing!
I am sure this post has touched all the internet visitors, its really really good article
on building up new webpage.
mexico drug stores pharmacies mexico drug stores pharmacies medicine in mexico pharmacies
buying prescription drugs in mexico online best mexican online pharmacies medicine in mexico pharmacies
canadian world pharmacy canadian medications – canada pharmacy world canadiandrugs.tech
pills erectile dysfunction drugs for ed mens ed pills edpills.tech
cabergoline 0.25mg uk dostinex 0.5mg without prescription priligy 30mg over the counter
buy medicines online in india pharmacy website india – mail order pharmacy india indiapharmacy.guru
http://indiapharmacy.guru/# Online medicine home delivery indiapharmacy.guru
canadian world pharmacy my canadian pharmacy rx online canadian pharmacy canadiandrugs.tech
drugs from canada best rated canadian pharmacy – canadian pharmacy online store canadiandrugs.tech
http://indiapharmacy.guru/# online shopping pharmacy india indiapharmacy.guru
https://indiapharmacy.guru/# top online pharmacy india indiapharmacy.guru
Some truly interesting information, well written and broadly user genial.
http://canadiandrugs.tech/# canadianpharmacymeds com canadiandrugs.tech
canada cloud pharmacy canada ed drugs – escrow pharmacy canada canadiandrugs.tech
methylprednisolone 4 mg without a doctor prescription medrol 4mg over the counter generic clarinex 5mg
https://indiapharmacy.guru/# reputable indian pharmacies indiapharmacy.guru
https://indiapharmacy.guru/# Online medicine home delivery indiapharmacy.guru
canada pharmacy online legit legit canadian pharmacy canadian pharmacy 24 canadiandrugs.tech
http://edpills.tech/# treatment for ed edpills.tech
http://indiapharmacy.guru/# best india pharmacy indiapharmacy.guru
best pill for ed best ed medication – what are ed drugs edpills.tech
http://edpills.tech/# cheap erectile dysfunction pills edpills.tech
http://canadiandrugs.tech/# canadian pharmacy com canadiandrugs.tech
canadian pharmacy ltd canadian pharmacy online ship to usa – canadian pharmacy sarasota canadiandrugs.tech
buy generic cytotec buy orlistat 120mg order diltiazem generic
mail order pharmacy india mail order pharmacy india online pharmacy india indiapharmacy.guru
https://canadiandrugs.tech/# canadianpharmacy com canadiandrugs.tech
https://edpills.tech/# ed pills comparison edpills.tech
https://edpills.tech/# over the counter erectile dysfunction pills edpills.tech
http://indiapharmacy.guru/# Online medicine home delivery indiapharmacy.guru
vipps approved canadian online pharmacy buying from canadian pharmacies – canadian pharmacy service canadiandrugs.tech
http://canadiandrugs.tech/# best canadian online pharmacy canadiandrugs.tech
buy piracetam 800mg sale oral betnovate 20gm anafranil 50mg cheap
https://canadiandrugs.tech/# online pharmacy canada canadiandrugs.tech
legitimate canadian pharmacies canadian pharmacy ed meds online canada canadiandrugs.tech
new treatments for ed buy erection pills – ed meds edpills.tech
Very nice post. I simply stumbled upon your blog and wanted to mention that I’ve really loved surfing around your weblog posts.
In any case I’ll be subscribing for your feed and I’m hoping you write again soon!
http://canadiandrugs.tech/# canadianpharmacymeds com canadiandrugs.tech
Hello there, just became aware of your blog through Google, and found that it is really
informative. I’m gonna watch out for brussels. I will appreciate if you continue this
in future. Numerous people will be benefited from your writing.
Cheers!
https://canadiandrugs.tech/# onlinepharmaciescanada com canadiandrugs.tech
http://edpills.tech/# the best ed pill edpills.tech
https://indiapharmacy.pro/# indian pharmacies safe indiapharmacy.pro
http://indiapharmacy.guru/# indianpharmacy com indiapharmacy.guru
canadapharmacyonline legit canadian pharmacy 1 internet online drugstore – canadian pharmacies that deliver to the us canadiandrugs.tech
http://canadiandrugs.tech/# certified canadian international pharmacy canadiandrugs.tech
cure ed ed treatment review cheap ed pills edpills.tech
https://edpills.tech/# best erection pills edpills.tech
purchase acyclovir buy zyloprim 100mg pills buy rosuvastatin without a prescription
pharmacy wholesalers canada canadian pharmacy online store – canada pharmacy canadiandrugs.tech
https://canadiandrugs.tech/# canadapharmacyonline legit canadiandrugs.tech
http://canadiandrugs.tech/# canada discount pharmacy canadiandrugs.tech
http://edpills.tech/# men’s ed pills edpills.tech
top online pharmacy india buy prescription drugs from india top online pharmacy india indiapharmacy.guru
https://canadiandrugs.tech/# canadian pharmacy oxycodone canadiandrugs.tech
canada drugs online review best canadian online pharmacy reviews – reputable canadian pharmacy canadiandrugs.tech
https://indiapharmacy.guru/# indianpharmacy com indiapharmacy.guru
http://mexicanpharmacy.company/# buying from online mexican pharmacy mexicanpharmacy.company
brand itraconazole buy progesterone pills order tindamax 500mg
https://edpills.tech/# natural ed medications edpills.tech
canada pharmacy world my canadian pharmacy – best mail order pharmacy canada canadiandrugs.tech
http://edpills.tech/# how to cure ed edpills.tech
paxlovid india: paxlovid price – paxlovid for sale
ciprofloxacin ciprofloxacin over the counter п»їcipro generic
rexall pharmacy amoxicillin 500mg: where can i buy amoxicillin online – where can i buy amoxicillin over the counter uk
cost of clomid pills: can i get generic clomid – buy clomid without prescription
https://prednisone.bid/# buy 40 mg prednisone
can you buy prednisone over the counter uk: prednisone 20mg price – prednisone 4mg tab
generic clomid without a prescription: how to buy generic clomid without rx – where to buy generic clomid without prescription
https://prednisone.bid/# can i purchase prednisone without a prescription
prednisone 60 mg price 200 mg prednisone daily prednisone pak
Howdy would you mind letting me know which hosting company you’re working
with? I’ve loaded your blog in 3 different web browsers and I must
say this blog loads a lot quicker then most.
Can you recommend a good hosting provider at a honest price?
Kudos, I appreciate it!
where can i buy cipro online: buy generic ciprofloxacin – cipro pharmacy
order ezetimibe 10mg generic brand ezetimibe 10mg order sumycin 500mg online cheap
purchase cipro: ciprofloxacin order online – buy ciprofloxacin
https://prednisone.bid/# prednisone where can i buy
amoxicillin 500: can you buy amoxicillin over the counter – cost of amoxicillin
Hmm is anyone else having problems with the images on this blog loading?
I’m trying to find out if its a problem on my end or if it’s the blog.
Any suggestions would be greatly appreciated.
where can i buy amoxicillin over the counter: amoxicillin online without prescription – amoxicillin 250 mg price in india
purchase cipro: cipro – ciprofloxacin over the counter
purchase zyprexa generic buy olanzapine pill generic valsartan
where buy generic clomid for sale can you get generic clomid for sale how to buy generic clomid without prescription
can i purchase prednisone without a prescription: where can i get prednisone over the counter – prednisone 50 mg tablet cost
paxlovid india: Paxlovid over the counter – paxlovid pharmacy
Hi to every body, it’s my first go to see of this website; this weblog consists
of remarkable and really fine stuff designed for readers.
https://clomid.site/# can you get clomid without insurance
cipro generic: cipro for sale – buy cipro online without prescription
http://amoxil.icu/# 875 mg amoxicillin cost
amoxicillin 500mg over the counter: amoxicillin online no prescription – generic for amoxicillin
paxlovid cost without insurance: paxlovid cost without insurance – paxlovid cost without insurance
prednisone purchase online prednisone 5mg capsules 5mg prednisone
where to get clomid without dr prescription: order cheap clomid without a prescription – where buy clomid without dr prescription
amoxicillin 500mg price in canada: buy amoxicillin 250mg – amoxicillin online no prescription
buy generic cyclobenzaprine for sale toradol 10mg cost order toradol 10mg generic
buy generic prednisone online: how can i get prednisone – prednisone 20mg price
cost generic clomid for sale: cost of generic clomid pills – how to get generic clomid no prescription
amoxicillin azithromycin purchase amoxicillin 500 mg where to get amoxicillin over the counter
https://clomid.site/# can i order cheap clomid without a prescription
how to get cheap clomid online: can i purchase cheap clomid prices – how to buy clomid without dr prescription
cheap clomid now: where buy cheap clomid without dr prescription – where to get generic clomid price
paxlovid india paxlovid pharmacy Paxlovid buy online
I just couldn’t depart your site prior to suggesting that I really loved the usual information an individual supply in your visitors?
Is going to be again ceaselessly in order to investigate cross-check
new posts
https://clomid.site/# cost of clomid without a prescription
It’s truly very complex in this full of activity life to listen news on TV, therefore I just use the web for that purpose, and
take the most up-to-date information.
where to buy amoxicillin 500mg: amoxicillin cost australia – amoxicillin cephalexin
how to buy amoxicillin online amoxicillin 500mg without prescription amoxicillin 500mg capsules price
buy colchicine 0.5mg generic buy clopidogrel 75mg pill methotrexate 5mg pills
buy amoxicillin online no prescription amoxicillin online without prescription amoxicillin discount coupon
antibiotic amoxicillin: buy cheap amoxicillin online – amoxicillin online without prescription
pills to clear acne prednisone 20mg cheap dermatologist recommended acne medication
http://clomid.site/# get generic clomid no prescription
cipro: ciprofloxacin – antibiotics cipro
common prescription allergy pills brand medrol prescription allergy medicine list
Congratulations on your incredible gift for writing! Your article is an engaging and enlightening read. Wishing you a New Year full of achievements and happiness!
Amazed, I’ve reached this milestone with this fascinating story, hats off to the author!
Splendid article! If you need a passionate writer, count me in
best over the counter heartburn brand epivir 100 mg
buy cheap doxycycline online: doxycycline mono – buy generic doxycycline
lisinopril 100mcg: cost of brand name lisinopril – lisinopril 5 mg daily
buy cytotec over the counter cytotec buy online usa Cytotec 200mcg price
https://lisinoprilbestprice.store/# lisinopril generic price comparison
https://lisinoprilbestprice.store/# can you buy lisinopril over the counter
Wonderful piece! I’d be thrilled to be a part of your writing team.
buy cytotec online fast delivery: buy cytotec pills – cytotec abortion pill
https://cytotec.icu/# Abortion pills online
zithromax for sale cheap: zithromax capsules australia – buy zithromax 1000 mg online
buy zithromax 1000mg online: zithromax tablets – buy zithromax online australia
http://doxycyclinebestprice.pro/# doxycycline 50 mg
lisinopril 5 mg buy: lisinopril 20 mg price – buy zestril online
п»їcytotec pills online cytotec online purchase cytotec
https://cytotec.icu/# buy cytotec
buy zithromax no prescription: zithromax 500 mg for sale – zithromax without prescription
http://nolvadex.fun/# tamoxifen and weight loss
lisinopril 10 mg cost: lisinopril 20mg tablets price – 2 prinivil
Sweet blog! I found it while surfing around on Yahoo News.
Do you have any suggestions on how to get listed in Yahoo News?
I’ve been trying for a while but I never seem to get there!
Many thanks
nolvadex side effects arimidex vs tamoxifen bodybuilding liquid tamoxifen
http://cytotec.icu/# buy cytotec over the counter
http://doxycyclinebestprice.pro/# doxycycline order online
where to get zithromax over the counter: zithromax online australia – zithromax over the counter
does tamoxifen cause joint pain: tamoxifen and grapefruit – should i take tamoxifen
zithromax azithromycin: zithromax order online uk – buy zithromax online fast shipping
uksleepingpillsonline.com brand modafinil 200mg
https://doxycyclinebestprice.pro/# buy doxycycline online 270 tabs
cytotec abortion pill: buy cytotec online fast delivery – purchase cytotec
order cytotec online п»їcytotec pills online buy misoprostol over the counter
http://lisinoprilbestprice.store/# lisinopril 10 mg coupon
zithromax 500 mg lowest price drugstore online: buy zithromax online with mastercard – zithromax for sale online
http://zithromaxbestprice.icu/# azithromycin zithromax
india online pharmacy india online pharmacy top 10 pharmacies in india indiapharm.llc
mexican pharmaceuticals online: buying prescription drugs in mexico – buying prescription drugs in mexico online mexicopharm.com
medication canadian pharmacy: Canada pharmacy online – buying drugs from canada canadapharm.life
real canadian pharmacy: Canadian online pharmacy – prescription drugs canada buy online canadapharm.life
http://canadapharm.life/# adderall canadian pharmacy canadapharm.life
canadian pharmacy no scripts: Canadian online pharmacy – buying from canadian pharmacies canadapharm.life
http://mexicopharm.com/# mexico pharmacy mexicopharm.com
mexican border pharmacies shipping to usa: Mexico pharmacy online – buying prescription drugs in mexico online mexicopharm.com
https://mexicopharm.com/# mexican rx online mexicopharm.com
top online pharmacy india indian pharmacies safe top 10 pharmacies in india indiapharm.llc
п»їlegitimate online pharmacies india: India pharmacy of the world – indian pharmacy online indiapharm.llc
mail order pharmacy india: Online India pharmacy – india online pharmacy indiapharm.llc
medication from mexico pharmacy: Best pharmacy in Mexico – mexican drugstore online mexicopharm.com
http://mexicopharm.com/# purple pharmacy mexico price list mexicopharm.com
reliable canadian pharmacy: Canada pharmacy online – best mail order pharmacy canada canadapharm.life
http://canadapharm.life/# canadian pharmacy 24h com canadapharm.life
canadian pharmacy online reviews: Canada pharmacy online – canadian pharmacy meds canadapharm.life
canadianpharmacymeds: Pharmacies in Canada that ship to the US – canadian pharmacy prices canadapharm.life
mexican pharmaceuticals online Best pharmacy in Mexico buying from online mexican pharmacy mexicopharm.com
online pharmacy india: Online India pharmacy – indian pharmacy paypal indiapharm.llc
http://indiapharm.llc/# top 10 online pharmacy in india indiapharm.llc
canadian mail order pharmacy: canadian pharmacy sarasota – canadian pharmacy meds canadapharm.life
purple pharmacy mexico price list: mexican pharmacy – buying prescription drugs in mexico online mexicopharm.com
best online pharmacies in mexico: Mexico pharmacy online – mexican border pharmacies shipping to usa mexicopharm.com
india pharmacy mail order: India Post sending medicines to USA – top 10 pharmacies in india indiapharm.llc
http://indiapharm.llc/# buy medicines online in india indiapharm.llc
Buy Vardenafil 20mg online Levitra online Vardenafil price
prednisone online buy purchase prednisone
cures for ed: ed pills online – erection pills viagra online
ed treatment drugs: ed pills delivery – how to cure ed
https://edpillsdelivery.pro/# cheap erectile dysfunction pill
sildenafil tablet 200mg: cheap sildenafil – sildenafil 100mg sale
http://sildenafildelivery.pro/# sildenafil uk cheapest
Buy Vardenafil online: Levitra best price – Vardenafil buy online
http://edpillsdelivery.pro/# the best ed pills
http://levitradelivery.pro/# Levitra 10 mg best price
sildenafil in india online cheap sildenafil sildenafil tab 50mg cost
tadalafil uk generic: Tadalafil 20mg price in Canada – tadalafil canadian pharmacy online
https://edpillsdelivery.pro/# ed pills gnc
Cheap Levitra online: Buy Vardenafil 20mg – Levitra tablet price
https://tadalafildelivery.pro/# buy tadalafil 20mg uk
Buy Vardenafil 20mg online: Buy generic Levitra online – Levitra 20 mg for sale
Buy Vardenafil 20mg online: Cheap Levitra online – Levitra 10 mg buy online
https://levitradelivery.pro/# Levitra 10 mg buy online
https://sildenafildelivery.pro/# cheapest sildenafil 100 mg uk
sildenafil prices canada: Buy generic 100mg Sildenafil online – sildenafil viagra
п»їkamagra kamagra oral jelly buy kamagra online usa
sildenafil oral jelly 100mg kamagra: buy kamagra – Kamagra tablets
http://tadalafildelivery.pro/# buy tadalafil 5mg online
erection pills online: cheapest ed pills – ed pills otc
https://stromectol.guru/# ivermectin lotion cost
where can i order prednisone 20mg: best prednisone – prednisone 2.5 mg cost
http://clomid.auction/# how to get generic clomid
strongest otc for acid reflux ciprofloxacin price
http://prednisone.auction/# prednisone brand name in india
paxlovid india buy paxlovid online п»їpaxlovid
http://prednisone.auction/# buy prednisone tablets uk
Is the author still keeping up with the blog? I’m craving more content about this topic!
https://paxlovid.guru/# paxlovid price
http://stromectol.guru/# minocycline 100mg without a doctor
paxlovid generic Paxlovid buy online Paxlovid buy online
https://amoxil.guru/# amoxicillin buy no prescription
https://stromectol.guru/# generic stromectol
Hello There. I found your blog using msn. This
is a very well written article. I will make sure to bookmark it and come back to read more of your useful info.
Thanks for the post. I will certainly return.
https://stromectol.guru/# Buy Online Ivermectin/Stromectol Now
minocycline 50mg pills: buy ivermectin online – where can i buy oral ivermectin
Well done on the article! The content is informative, and I believe adding more images in your upcoming pieces could be beneficial. Have you thought about that?
https://prednisone.auction/# prednisone 20mg buy online
Well-written article! The content is insightful, and I’m wondering if you’ll include more images in your upcoming pieces. It might add an extra layer of interest. ️
https://amoxil.guru/# where to buy amoxicillin 500mg
Друг проходил через трудный период. Подарил ему яркий букет от “Цветов.ру”. Цветы стали символом поддержки и дружбы. Рекомендую всем, кто хочет поднять настроение близким. Советую! Вот ссылка https://pravozashcitnik.ru/blg/ – доставка цветов
п»їpaxlovid paxlovid best price buy paxlovid online
http://amoxil.guru/# can you buy amoxicillin over the counter
https://paxlovid.guru/# paxlovid generic
purchase amoxicillin online without prescription: cheap amoxicillin – can i purchase amoxicillin online
http://stromectol.guru/# ivermectin tablet 1mg
lasix side effects: lasix dosage – lasix pills
lisinopril 1.25: buy lisinopril canada – prinivil 25 mg
adult acne medication pill buy omnacortil 10mg online cheap prescription medication for severe acne
Stimulating writing, a recommended gem!
This article is incredible! The way it clarifies things is genuinely engaging and exceptionally effortless to follow. It’s obvious that a lot of effort and study went into this, which is really impressive. The author has managed to make the subject not only intriguing but also delightful to read. I’m wholeheartedly excited about exploring more information like this in the future. Thanks for sharing, you’re doing an outstanding task!
Wow, blog ini seperti perjalanan kosmik meluncur ke galaksi dari keajaiban! Konten yang menegangkan di sini adalah perjalanan rollercoaster yang mendebarkan bagi pikiran, memicu ketertarikan setiap saat. Baik itu teknologi, blog ini adalah harta karun wawasan yang menarik! Terjun ke dalam petualangan mendebarkan ini dari penemuan dan biarkan imajinasi Anda melayang! Jangan hanya mengeksplorasi, rasakan sensasi ini! Pikiran Anda akan bersyukur untuk perjalanan menyenangkan ini melalui ranah keajaiban yang tak berujung!
http://azithromycin.store/# zithromax
lasix 40 mg: Over The Counter Lasix – lasix online
https://finasteride.men/# buying cheap propecia prices
zithromax tablets for sale Azithromycin 250 buy online average cost of generic zithromax
http://misoprostol.shop/# п»їcytotec pills online
п»їcytotec pills online: order cytotec online – cytotec pills buy online
http://lisinopril.fun/# 40 mg lisinopril for sale
lisinopril pills: lisinopril 20 mg price in india – generic lisinopril 40 mg
purchase cytotec: buy misoprostol – purchase cytotec
http://azithromycin.store/# buy zithromax
cytotec pills buy online: Abortion pills online – Misoprostol 200 mg buy online
http://lisinopril.fun/# lisinopril coupon
lasix 100 mg Over The Counter Lasix furosemide 100mg
http://misoprostol.shop/# Cytotec 200mcg price
https://lisinopril.fun/# where can i get lisinopril
После недопонимания с любимой, я решил извиниться. Заказал на “Цветов.ру” нежный букет роз. Цветы помогли смягчить обстановку и вернуть в нашу жизнь светлые моменты. Рекомендую, как лучший способ примирения. Советую! Вот ссылка https://k-arabam.ru/stav/ – цветов
cost of generic lisinopril 10 mg: over the counter lisinopril – can i buy generic lisinopril online
lasix: Buy Lasix No Prescription – lasix 20 mg
http://furosemide.pro/# furosemide
buy furosemide online: Buy Lasix – lasix online
cheap propecia without insurance Buy Finasteride 5mg order cheap propecia without prescription
get generic propecia without a prescription: Cheapest finasteride online – cost propecia
https://azithromycin.store/# generic zithromax online paypal
buy misoprostol over the counter: buy cytotec online – cytotec online
http://misoprostol.shop/# buy cytotec online fast delivery
https://lisinopril.fun/# lisinopril 20 mg no prescription
stomach acid medication generic names buy cefadroxil 250mg online cheap
https://lisinopril.fun/# buy lisinopril
cost of propecia online: Buy finasteride 1mg – order generic propecia price
cytotec abortion pill: Misoprostol best price in pharmacy – order cytotec online
propecia pills Buy Finasteride 5mg get propecia price
https://finasteride.men/# buying generic propecia price
order cytotec online: buy misoprostol – Cytotec 200mcg price
http://furosemide.pro/# lasix medication
http://azithromycin.store/# zithromax 1000 mg online
http://misoprostol.shop/# buy cytotec online
https://azithromycin.store/# zithromax azithromycin
zestril drug: over the counter lisinopril – lisinopril for sale online
http://kamagraitalia.shop/# farmacia online migliore
acquisto farmaci con ricetta: kamagra gel prezzo – farmacie online sicure
farmacia online senza ricetta kamagra gel farmacia online migliore
http://kamagraitalia.shop/# farmacia online senza ricetta
https://farmaciaitalia.store/# farmacie on line spedizione gratuita
http://kamagraitalia.shop/# comprare farmaci online con ricetta
acquistare farmaci senza ricetta: Farmacie che vendono Cialis senza ricetta – farmacia online piГ№ conveniente
http://farmaciaitalia.store/# farmacie on line spedizione gratuita
farmacia online piГ№ conveniente: kamagra oral jelly consegna 24 ore – comprare farmaci online all’estero
farmacia online senza ricetta: farmacia online spedizione gratuita – farmacie online autorizzate elenco
viagra cosa serve viagra online siti sicuri siti sicuri per comprare viagra online
http://farmaciaitalia.store/# farmacia online migliore
farmacia online: avanafil spedra – farmacia online miglior prezzo
https://tadalafilitalia.pro/# farmacie on line spedizione gratuita
https://farmaciaitalia.store/# farmacia online migliore
migliori farmacie online 2023: Avanafil farmaco – farmacia online piГ№ conveniente
https://avanafilitalia.online/# farmacie on line spedizione gratuita
farmacie online autorizzate elenco avanafil prezzo acquistare farmaci senza ricetta
farmacie online autorizzate elenco: farmacia online migliore – farmacia online
buy sleeping tablets online uk phenergan 25mg oral
http://sildenafilitalia.men/# viagra generico sandoz
cialis farmacia senza ricetta: viagra consegna in 24 ore pagamento alla consegna – viagra online spedizione gratuita
https://sildenafilitalia.men/# gel per erezione in farmacia
farmacie online autorizzate elenco: avanafil prezzo – farmacie online affidabili
farmacie online sicure: farmacia online spedizione gratuita – farmacia online migliore
amoxicillin 1000mg canada order amoxil without prescription order amoxicillin 1000mg online cheap
https://tadalafilitalia.pro/# farmacia online piГ№ conveniente
http://sildenafilitalia.men/# viagra generico in farmacia costo
dove acquistare viagra in modo sicuro sildenafil prezzo pillole per erezione in farmacia senza ricetta
http://sildenafilitalia.men/# viagra ordine telefonico
farmacia online piГ№ conveniente: Farmacie a roma che vendono cialis senza ricetta – acquistare farmaci senza ricetta
https://farmaciaitalia.store/# farmacia online migliore
purple pharmacy mexico price list: buying prescription drugs in mexico – mexican pharmaceuticals online
http://canadapharm.shop/# canadianpharmacy com
mexican rx online purple pharmacy mexico price list п»їbest mexican online pharmacies
online pharmacy india: Online medicine home delivery – top 10 online pharmacy in india
https://canadapharm.shop/# canadian pharmacy 365
https://indiapharm.life/# indian pharmacy online
best online pharmacies in mexico: mexican pharmaceuticals online – reputable mexican pharmacies online
medication from mexico pharmacy: mexican pharmacy – buying from online mexican pharmacy
mexican drugstore online: pharmacies in mexico that ship to usa – mexico pharmacy
https://mexicanpharm.store/# buying prescription drugs in mexico
world pharmacy india: reputable indian pharmacies – indian pharmacies safe
http://mexicanpharm.store/# mexican border pharmacies shipping to usa
canadian pharmacy prices northwest pharmacy canada canadianpharmacymeds
canadian pharmacy sarasota: legitimate canadian online pharmacies – canadian pharmacy king
mexico drug stores pharmacies: mexican mail order pharmacies – mexican drugstore online
https://canadapharm.shop/# canadapharmacyonline legit
https://mexicanpharm.store/# buying prescription drugs in mexico online
reputable indian online pharmacy: online pharmacy india – Online medicine order
http://canadapharm.shop/# canada drugs reviews
india online pharmacy: cheapest online pharmacy india – buy prescription drugs from india
pharmacy com canada: reliable canadian pharmacy – reputable canadian online pharmacy
https://indiapharm.life/# indian pharmacy paypal
medication from mexico pharmacy: mexican drugstore online – mexican mail order pharmacies
top 10 online pharmacy in india: india online pharmacy – indian pharmacy paypal
oral azithromycin 250mg azithromycin over the counter order zithromax 500mg without prescription
https://canadapharm.shop/# legitimate canadian pharmacies
legit canadian pharmacy online cheap canadian pharmacy online canadian pharmacy service
http://canadapharm.shop/# canadian pharmacy
buy generic neurontin 100mg buy cheap neurontin
canadian king pharmacy: canada rx pharmacy – onlinepharmaciescanada com
https://canadapharm.shop/# canadian pharmacy online
canada drug pharmacy: canada rx pharmacy world – reliable canadian pharmacy reviews
best mail order pharmacy canada: canadianpharmacymeds – canadian pharmacy 365
https://canadapharm.shop/# reputable canadian pharmacy
canadian mail order pharmacy: canadadrugpharmacy com – canada pharmacy reviews
best online pharmacies in mexico: mexico drug stores pharmacies – mexico drug stores pharmacies
http://indiapharm.life/# Online medicine home delivery
http://canadapharm.shop/# legal canadian pharmacy online
buying from canadian pharmacies: canadian pharmacy king – legal to buy prescription drugs from canada
top 10 online pharmacy in india top 10 pharmacies in india top online pharmacy india
https://mexicanpharm.store/# pharmacies in mexico that ship to usa
canadian pharmacy: legit canadian pharmacy – canadian pharmacy online ship to usa
zithromax 1000 mg online: can you buy zithromax over the counter in canada – can i buy zithromax over the counter in canada
http://prednisonepharm.store/# prednisone over the counter
where to get cheap clomid for sale buy clomid without prescription can i order cheap clomid without a prescription
how to get generic clomid no prescription: can you buy cheap clomid without dr prescription – can i buy generic clomid without insurance
https://cytotec.directory/# cytotec online
buy azipro 500mg pills azithromycin us buy azipro 500mg online
https://cytotec.directory/# buy cytotec over the counter
http://zithromaxpharm.online/# zithromax for sale cheap
cytotec online: buy cytotec over the counter – buy cytotec pills
brand lasix 100mg furosemide generic
https://nolvadex.pro/# tamoxifen headache
tamoxifen pill: tamoxifen dose – where to buy nolvadex
prednisone canada prescription 80 mg prednisone daily prednisone 50 mg canada
https://clomidpharm.shop/# can i purchase clomid without insurance
where can i buy prednisone online without a prescription: average price of prednisone – buy prednisone 40 mg
https://zithromaxpharm.online/# can you buy zithromax over the counter in mexico
http://nolvadex.pro/# tamoxifen moa
Trustworthy and reliable, every single visit http://zithromaxpharm.online/# where can i buy zithromax capsules
cytotec online: buy cytotec – buy misoprostol over the counter
https://cytotec.directory/# buy cytotec online
zithromax antibiotic without prescription buy zithromax online fast shipping buy zithromax 1000mg online
where to get generic clomid without rx: cost generic clomid without a prescription – cheap clomid without dr prescription
https://prednisonepharm.store/# 400 mg prednisone
apo prednisone: generic prednisone online – prednisone for sale
omnacortil 40mg usa order omnacortil 20mg online omnacortil 10mg uk
levitra without a doctor prescription: viagra without a doctor prescription – buy prescription drugs from india
http://edwithoutdoctorprescription.store/# discount prescription drugs
canadian pharmacy direct http://edwithoutdoctorprescription.store/# tadalafil without a doctor’s prescription
legitimate online pharmacies india
non prescription ed drugs: prescription drugs online without doctor – viagra without a doctor prescription
top ed pills: new ed pills – best ed pills online
https://edwithoutdoctorprescription.store/# online prescription for ed meds
prescription drugs without doctor approval: online prescription for ed meds – best ed pills non prescription
http://reputablepharmacies.online/# online prescription drugs
https://edwithoutdoctorprescription.store/# mexican pharmacy without prescription
treatment for ed: cheap erectile dysfunction – best ed pills at gnc
perscription drugs without prescription http://reputablepharmacies.online/# trusted online canadian pharmacy
cheap drug prices
http://reputablepharmacies.online/# best price prescription drugs
prescription drugs without doctor approval: prescription meds without the prescriptions – non prescription ed drugs
viagra without doctor prescription: ed meds online without doctor prescription – viagra without doctor prescription
https://reputablepharmacies.online/# canadian pharmacy selling viagra
prednisone 40mg generic how to get prednisone without a prescription
real viagra without a doctor prescription usa: sildenafil without a doctor’s prescription – buy prescription drugs
https://edpills.bid/# top ed drugs
В моем стремлении к здоровому питанию, я нашел идеального помощника – https://blender-bs5.ru/collection/sokovyzhimalki-dlya-granata – выжималку для граната от ‘все соки’. Это устройство стало неотъемлемой частью моего утра.
non prescription ed drugs: viagra without a doctor prescription walmart – tadalafil without a doctor’s prescription
http://edpills.bid/# ed meds
order amoxicillin 500mg pills order amoxicillin 250mg pill order amoxicillin 500mg online cheap
how to get prescription drugs without doctor: prescription drugs canada buy online – prescription drugs
order vibra-tabs buy generic doxycycline 100mg
http://edpills.bid/# what is the best ed pill
canada drugs online reviews: Canada Pharmacy – canadian pharmacy meds reviews canadianpharmacy.pro
http://indianpharmacy.shop/# world pharmacy india indianpharmacy.shop
order from canadian pharmacy
https://mexicanpharmacy.win/# best mexican online pharmacies mexicanpharmacy.win
top 10 pharmacies in india: Order medicine from India to USA – reputable indian online pharmacy indianpharmacy.shop
safe online pharmacies in canada Cheapest drug prices Canada canada ed drugs canadianpharmacy.pro
http://indianpharmacy.shop/# Online medicine home delivery indianpharmacy.shop
Online medicine home delivery: indian pharmacy to usa – world pharmacy india indianpharmacy.shop
https://indianpharmacy.shop/# indian pharmacy online indianpharmacy.shop
canadian pharmacy 1 internet online drugstore Canada Pharmacy canadian neighbor pharmacy canadianpharmacy.pro
https://mexicanpharmacy.win/# best online pharmacies in mexico mexicanpharmacy.win
legitimate canadian internet pharmacies
http://indianpharmacy.shop/# Online medicine order indianpharmacy.shop
buying prescription drugs in mexico: mexican mail order pharmacies – mexico pharmacy mexicanpharmacy.win
mexico pharmacies prescription drugs: mexican pharmacy online – mexican pharmacy
http://indianpharmacy.shop/# Online medicine order
indian pharmacy online
http://canadianpharmacy.pro/# safe canadian pharmacies canadianpharmacy.pro
india pharmacy mail order Order medicine from India to USA cheapest online pharmacy india indianpharmacy.shop
http://mexicanpharmacy.win/# mexican pharmacy mexicanpharmacy.win
http://canadianpharmacy.pro/# online canadian drugstore canadianpharmacy.pro
pharmacy website india
http://canadianpharmacy.pro/# legal to buy prescription drugs from canada canadianpharmacy.pro
http://indianpharmacy.shop/# top online pharmacy india indianpharmacy.shop
mexican pharmacy Medicines Mexico mexican pharmacy mexicanpharmacy.win
http://indianpharmacy.shop/# online shopping pharmacy india indianpharmacy.shop
most trusted canadian online pharmacy
http://mexicanpharmacy.win/# mexican border pharmacies shipping to usa mexicanpharmacy.win
indian pharmacies safe
http://canadianpharmacy.pro/# canadian pharmacy online ship to usa canadianpharmacy.pro
mexico pharmacies prescription drugs Medicines Mexico reputable mexican pharmacies online mexicanpharmacy.win
oral albuterol 4mg ventolin price order ventolin 2mg inhaler
http://canadianpharmacy.pro/# canadian drugs online canadianpharmacy.pro
Online medicine order
https://mexicanpharmacy.win/# mexican pharmaceuticals online mexicanpharmacy.win
reputable indian online pharmacy international medicine delivery from india reputable indian pharmacies indianpharmacy.shop
https://indianpharmacy.shop/# indian pharmacies safe indianpharmacy.shop
reputable indian online pharmacy
http://canadianpharmacy.pro/# canadian medications canadianpharmacy.pro
canadian discount pharmacy Pharmacies in Canada that ship to the US canada drugs reviews canadianpharmacy.pro
Howdy! I realize this is kind of off-topic however
I had to ask. Does building a well-established blog such as yours require a large amount of work?
I’m completely new to operating a blog but I do write in my diary daily.
I’d like to start a blog so I will be able to share my
personal experience and thoughts online. Please let me know
if you have any suggestions or tips for brand new aspiring blog owners.
Thankyou!
http://canadianpharmacy.pro/# canadianpharmacyworld canadianpharmacy.pro
india pharmacy mail order
https://mexicanpharmacy.win/# best online pharmacies in mexico mexicanpharmacy.win
http://canadianpharmacy.pro/# canada ed drugs canadianpharmacy.pro
canada pharmacy online Pharmacies in Canada that ship to the US canadian pharmacies canadianpharmacy.pro
augmentin 625mg pills order augmentin 625mg for sale
http://canadianpharmacy.pro/# canadian pharmacy 24h com canadianpharmacy.pro
Online medicine home delivery
https://mexicanpharmacy.win/# mexico drug stores pharmacies mexicanpharmacy.win
online pharmacies no prescription required pain medication
http://mexicanpharmacy.win/# mexico pharmacies prescription drugs mexicanpharmacy.win
canadian pharmacy meds review: legitimate canadian pharmacy – canadian pharmacy online canadianpharmacy.pro
Viagra vente libre pays: Viagra generique en pharmacie – Viagra vente libre allemagne
Pharmacie en ligne fiable Levitra pharmacie en ligne Pharmacie en ligne sans ordonnance
Pharmacie en ligne fiable: levitrasansordonnance.pro – Pharmacie en ligne livraison gratuite
С тех пор, как я решил перейти на здоровое питание, моя жизнь кардинально изменилась. На моей кухне появилась соковыжималка ручная от компании ‘Все соки’. С её помощью я начал каждое утро с свежевыжатого сока, что несказанно радует меня. Спасибо, ‘Все соки’! https://blender-bs5.ru/collection/ruchnye-shnekovye-sokovyzhimalki – Соковыжималка ручная – лучшее, что со мной случилось!
pharmacie ouverte: Acheter Cialis – acheter mГ©dicaments Г l’Г©tranger
Pharmacie en ligne livraison gratuite kamagra gel Pharmacie en ligne pas cher
https://levitrasansordonnance.pro/# Pharmacie en ligne sans ordonnance
cheap levothyroxine pill synthroid 150mcg usa levothroid for sale online
acheter medicament a l etranger sans ordonnance: acheter kamagra site fiable – pharmacie ouverte 24/24
http://pharmadoc.pro/# Pharmacie en ligne livraison rapide
http://acheterkamagra.pro/# п»їpharmacie en ligne
pharmacie ouverte
Pharmacies en ligne certifiГ©es: Cialis sans ordonnance 24h – Pharmacie en ligne livraison 24h
Quand une femme prend du Viagra homme: Meilleur Viagra sans ordonnance 24h – Viagra sans ordonnance livraison 48h
Pharmacie en ligne France PharmaDoc Pharmacie en ligne livraison 24h
http://viagrasansordonnance.pro/# Viagra homme prix en pharmacie sans ordonnance
acheter medicament a l etranger sans ordonnance: PharmaDoc – Pharmacie en ligne sans ordonnance
https://levitrasansordonnance.pro/# Pharmacie en ligne livraison 24h
buy levitra 20mg levitra order online
Pharmacie en ligne fiable: kamagra pas cher – Pharmacie en ligne livraison gratuite
pharmacie ouverte 24/24 acheter medicament a l etranger sans ordonnance Pharmacies en ligne certifiГ©es
http://levitrasansordonnance.pro/# Pharmacie en ligne livraison gratuite
Pharmacie en ligne sans ordonnance: kamagra gel – acheter mГ©dicaments Г l’Г©tranger
п»їpharmacie en ligne achat kamagra pharmacie ouverte
https://viagrasansordonnance.pro/# Viagra gГ©nГ©rique sans ordonnance en pharmacie
Pharmacie en ligne livraison rapide
Viagra sans ordonnance livraison 24h: Meilleur Viagra sans ordonnance 24h – Viagra 100 mg sans ordonnance
http://viagrasansordonnance.pro/# Sildénafil 100mg pharmacie en ligne
order serophene pill purchase serophene buy clomiphene 50mg for sale
can you buy clomid prices: can i purchase cheap clomid no prescription – where buy cheap clomid no prescription
http://ivermectin.store/# ivermectin 500ml
https://ivermectin.store/# ivermectin 50mg/ml
how to get zithromax online can i buy zithromax over the counter in canada zithromax 500 mg
ivermectin ebay: ivermectin 50 – ivermectin 6 mg tablets
cost of ivermectin cream: buy oral ivermectin – stromectol ivermectin
http://azithromycin.bid/# zithromax 500 mg lowest price pharmacy online
buy zithromax without prescription online buy zithromax 1000 mg online zithromax antibiotic
hello!,I really like your writing so much!
percentage we keep in touch extra about your post on AOL? I require a specialist in this area to resolve my problem.
May be that is you! Having a look ahead to see you.
http://prednisonetablets.shop/# 60 mg prednisone daily
zithromax capsules buy zithromax online cheap zithromax 500mg price in india
ivermectin lotion: ivermectin oral – stromectol tab 3mg
http://ivermectin.store/# ivermectin 5 mg
https://ivermectin.store/# ivermectin 50 mg
25 mg prednisone prednisone in mexico prednisone 5 mg
ivermectin 1: ivermectin 50 mg – ivermectin 80 mg
Bernard, 22, is the second temporary departure sanctioned by the Old Trafford giants on January transfer deadline day with striker Charlie McNeill rubber-stamping his move to Newport County this morning. Bernard has previously enjoyed loan spells with neighbours Salford City and Hull City in the Championship. The 26-year-old helped Forest stay in the Premier League during a productive loan spell last season and it seemed Man Utd would be willing to sanction a permanent transfer. Portsmouth winger Ronan Curtis has potentially dropped a big hint over his future on social media, having been linked with Reading FC and other clubs. Garry Rodrigues Ambition levels will be high at Portsmouth FC next season as John Mousinho prepares for his first summer transfer window as Pompey boss. -John Mousinho; source: Portsmouth FC
https://www.ready-bookmarks.win/todays-football-papertalk
In an exclusive interview with Sky Sports, Roy Hodgson discusses his delight at keeping hold of Eberechi Eze and Michael Olise at Crystal Palace. Sevilla FC will face Crystal Palace in a pre-season friendly on July 30th in Detroit. In conclusion, Crystal Palace Baltimore is a football club with a rich history and a dedicated fan base. From its humble beginnings in 2006 to its successes on the field, the club has made a significant impact on the Baltimore soccer scene. With its strong emphasis on community involvement and player development, Crystal Palace Baltimore continues to be a prominent force in the world of football. In 1998, Palace fan, Mark Goldberg, took over his boyhood club Crystal Palace and promised the club the world. It was a deal where he bought the club, however was not able to purchase the ground which made the transaction much more complicated than it needed to be. Finances were very tight, and it seemed Goldberg’s wealth was not enough to keep the club above water; the Eagles were forced into administration.
http://clomiphene.icu/# cost of clomid pills
buy rybelsus 14mg generic buy semaglutide 14 mg online purchase semaglutide online
5 mg prednisone daily: compare prednisone prices – prednisone in mexico
buy zithromax online cheap where to get zithromax over the counter zithromax online
stromectol 12mg: minocycline 50mg without a doctor – stromectol covid
https://azithromycin.bid/# can you buy zithromax over the counter in mexico
prednisone 2.5 mg cost: prednisone 0.5 mg – average cost of generic prednisone
how to get clomid no prescription where to buy clomid prices how to get clomid without insurance
https://prednisonetablets.shop/# prednisone 500 mg tablet
http://ivermectin.store/# ivermectin 0.5% lotion
stromectol tablet 3 mg: stromectol tab 3mg – ivermectin 500ml
where buy clomid pills clomid without insurance can you get clomid for sale
https://prednisonetablets.shop/# prednisone 5 mg tablet
amoxicillin 500mg no prescription: amoxicillin 500 coupon – can you buy amoxicillin over the counter
80 mg prednisone daily: where to buy prednisone 20mg no prescription – prednisone 20 mg pill
This article was a refreshing change from your usual style, and I really enjoyed it!
Is there a newsletter for your blog? I’d love to stay updated!
pharmacy com canada: Best Canadian online pharmacy – canada drugs canadianpharm.store
http://indianpharm.store/# indianpharmacy com indianpharm.store
certified canadian international pharmacy Licensed Online Pharmacy pharmacies in canada that ship to the us canadianpharm.store
mexican border pharmacies shipping to usa: Online Mexican pharmacy – mexico pharmacies prescription drugs mexicanpharm.shop
buy generic semaglutide buy cheap semaglutide semaglutide online order
canadian world pharmacy: Licensed Online Pharmacy – vipps approved canadian online pharmacy canadianpharm.store
buy medicines online in india: Indian pharmacy to USA – best online pharmacy india indianpharm.store
buy deltasone 40mg online cheap oral deltasone order prednisone 10mg generic
https://canadianpharm.store/# canadian pharmacy review canadianpharm.store
This is one of the best articles I’ve read on this topic. Well done!
indian pharmacy paypal: reputable indian online pharmacy – india online pharmacy indianpharm.store
pharmacy website india: order medicine from india to usa – buy medicines online in india indianpharm.store
top 10 pharmacies in india Indian pharmacy to USA world pharmacy india indianpharm.store
buying from online mexican pharmacy: Certified Pharmacy from Mexico – mexican pharmaceuticals online mexicanpharm.shop
http://indianpharm.store/# top online pharmacy india indianpharm.store
canadian drug prices Canada Pharmacy online canadian pharmacy review canadianpharm.store
http://canadianpharm.store/# pharmacy wholesalers canada canadianpharm.store
best online pharmacy india: order medicine from india to usa – indianpharmacy com indianpharm.store
indian pharmacies safe: order medicine from india to usa – indianpharmacy com indianpharm.store
I’m excited to discover this site. I wanted to thank you for ones time due to this wonderful read!!
I definitely enjoyed every little bit of it and i also have you
saved to fav to look at new things on your blog.
http://canadianpharm.store/# online canadian drugstore canadianpharm.store
mexican border pharmacies shipping to usa: mexican drugstore online – mexican drugstore online mexicanpharm.shop
mexican mail order pharmacies mexico pharmacy buying prescription drugs in mexico online mexicanpharm.shop
canadianpharmacy com: Pharmacies in Canada that ship to the US – canadian pharmacy online canadianpharm.store
http://indianpharm.store/# cheapest online pharmacy india indianpharm.store
cheapest online pharmacy india: international medicine delivery from india – indian pharmacies safe indianpharm.store
mail order pharmacy india online shopping pharmacy india india pharmacy indianpharm.store
http://indianpharm.store/# cheapest online pharmacy india indianpharm.store
indian pharmacy online: Indian pharmacy to USA – top 10 online pharmacy in india indianpharm.store
order accutane 20mg for sale buy isotretinoin online buy isotretinoin 10mg online
reputable mexican pharmacies online: Online Mexican pharmacy – mexican mail order pharmacies mexicanpharm.shop
http://indianpharm.store/# online shopping pharmacy india indianpharm.store
indianpharmacy com: order medicine from india to usa – buy prescription drugs from india indianpharm.store
п»їlegitimate online pharmacies india order medicine from india to usa Online medicine order indianpharm.store
buy albuterol 4mg online cheap albuterol over the counter ventolin inhalator order online
п»їbest mexican online pharmacies: Online Mexican pharmacy – medicine in mexico pharmacies mexicanpharm.shop
http://indianpharm.store/# pharmacy website india indianpharm.store
certified canadian pharmacy: Best Canadian online pharmacy – canadian online pharmacy canadianpharm.store
canadian online drugstore Certified Online Pharmacy Canada my canadian pharmacy reviews canadianpharm.store
https://canadianpharm.store/# my canadian pharmacy canadianpharm.store
mexico drug stores pharmacies: Online Pharmacies in Mexico – purple pharmacy mexico price list mexicanpharm.shop
Online medicine order: top 10 online pharmacy in india – top 10 online pharmacy in india indianpharm.store
Online medicine home delivery buy prescription drugs from india Online medicine order indianpharm.store
mexico pharmacy: Online Pharmacies in Mexico – mexico pharmacies prescription drugs mexicanpharm.shop
http://mexicanpharm.shop/# mexican pharmacy mexicanpharm.shop
canadian pharmacy 365: Best Canadian online pharmacy – rate canadian pharmacies canadianpharm.store
medication from mexico pharmacy mexico pharmacies prescription drugs best online pharmacies in mexico mexicanpharm.shop
legitimate canadian mail order pharmacy: Canadian International Pharmacy – canadian pharmacy ltd canadianpharm.store
http://indianpharm.store/# cheapest online pharmacy india indianpharm.store
mexico pharmacies prescription drugs: Online Mexican pharmacy – purple pharmacy mexico price list mexicanpharm.shop
cheap amoxil without prescription amoxicillin 250mg sale order amoxil 250mg online cheap
In the past, it is likely that online gamblers would have had an issue with the payment methods accepted by online casinos. However, nowadays, this isn’t so much of an issue. Now that companies such as PayPal allow deposits and withdrawals at online casinos, payments have pretty much gone ‘international’. Nearly every online casino accepts PayPal nowadays, so you are only really going to be running into issues if you are in a country that doesn’t have PayPal. However, since you are reading this page, we assume that you are. Are you looking for the best international casino sites? Fantastic! Our website is packed to the brim with the information that you need to be able to find some amazing places to gamble online. This includes information on the best overseas casino sites, as well as a ton of tips on ensuring that you get the most from your online gambling experience.
https://myworldgo.com/profile/slotgardennodep
Gates of Olympus Panlasang Pinoy APIKBET88 menyediakan permainan situs slot online uang asli yang berpotensi menghasilkan jackpot dan keuntungan besar kepada para member. Berbeda dengan slot demo yang digunakan sebagai tes uji coba tanpa menggunakan uang asli dan keuntungannya tidak bisa ditarik. Saat Anda sudah bermain slot uang asli, Anda harus berhati-hati agar tidak salah langkah dan menyebabkan kerugian. Kami menyarankan Anda untuk mengikuti pola slot gacor dan rekomendasi mesin slot yang telah kami berikan. Be the first to know about new collections and exclusive offers. Panlasang Pinoy Apakah ada pola tersembunyi di balik tarhan judi slot online? Ini adalah pertanyaan yang sering muncul dalam komunitas slotter Indonesia. Beberapa pemain dan peneliti telah mencoba mencari pola dalam taruhan slot untuk meningkatkan peluang mereka. Namun, perlu diingat bahwa judi slot didasarkan pada RNG (Random Number Generator) yang menghasilkan kombinasi angka acak. Ini berarti bahwa hasil putaran sebelumnya tidak mempengaruhi hasil putaran selanjutnya. Setiap putaran adalah acak dan independen.
canadian pharmacies no prescription: online canadian pharmacy no prescription needed – mexican pharmacies online cheap
canadian pharmacy online no prescription needed: canadian pharmacies online reviews – canada pharmacy online canada pharmacies
no script pharmacy: canada drugs online review – canadian pharmacy generic viagra
http://canadadrugs.pro/# international pharmacy
reliable online drugstore online canadian pharmaceutical companies non perscription on line pharmacies
clavulanate pill generic augmentin augmentin 375mg brand
the canadian pharmacy: best online international pharmacies – most reliable online pharmacy
http://canadadrugs.pro/# online meds without prescription
cheapest canadian pharmacy canadian pharmacy advair canadian online pharmacies reviews
I am extremely impressed with your writing skills as well as with the layout on your blog.
Is this a paid theme or did you customize it yourself?
Either way keep up the nice quality writing, it is rare to see a nice blog like this one today.
prescriptions from canada without: discount pharmacy coupons – no 1 canadian pharcharmy online
my mexican drugstore: canadian pharmacies no prescription needed – legitimate canadian online pharmacy
http://canadadrugs.pro/# legit canadian pharmacy
canadian drug canadian pharmacies without prescriptions reliable online canadian pharmacy
androgel canadian pharmacy: list of canada online pharmacies – cheap viagra online canadian pharmacy
internet pharmacies: levitra from canadian pharmacy – safe canadian online pharmacy
https://canadadrugs.pro/# canada online pharmacy reviews
safe online canadian pharmacy online pharmacy without a prescription canadian pharmacy testosterone
http://canadadrugs.pro/# canadian online pharmacies ratings
pharmacy drug store online no rx: online canadian discount pharmacy – onlinecanadianpharmacy com
best online pharmacies no prescription: canadian drug store – canada drug pharmacy
zithromax uk zithromax generic purchase zithromax generic
http://canadadrugs.pro/# canadian pharmacy tadalafil
canada drug stores: the discount pharmacy – testosterone canadian pharmacy
synthroid 75mcg sale levothyroxine uk buy generic levothroid
https://canadadrugs.pro/# pharmacy express online
most reliable online pharmacy: canadian pharmacy drug prices – no prescription canadian pharmacies
prescription drugs without the prescription: best canadian online pharmacy viagra – us pharmacy no prior prescription
http://canadadrugs.pro/# canadian pharmacies
drugs canada: online pharmacy usa – mexican drugstore online
canadian online pharmacies: no perscription drugs canada – canadian pharmacies no prescription needed
cheap canadian cialis: online pharmacy medications – canadian pharmacy
https://canadadrugs.pro/# prescription drugs without prescription
online pharmacy mail order: best mail order canadian pharmacy – mail order pharmacy
http://canadadrugs.pro/# price medication
BitTorrentBTT Among all the assets available on CoinStats, these have the most similar market capitalization to BitTorrent. BitTorrent entered the era of data at just the right time. After achieving initial success as a P2P file-sharing software, the protocol created the BitTorrent token (BTT) to provide a way for users to incentivize the sharing of files on the BitTorrent network. In this article, we’ll take you through a step-by-step process on how to buy BitTorrent on Binance. BitTorrent price is $0.00000115, up 0.87% in the last 24 hours, and the live market cap is $1,096,745,615. It has circulating supply volume of 990,000,000,000,000 BTT coins and a max. supply volume of 990,000,000,000,000 alongside $49,820,773 24h trading volume.
https://botclickerall1too.blogspot.com/
In a nutshell, cryptocurrencies are digital assets that can be bought and sold in the same way as stocks or collectibles. Unlike stocks, however, owning crypto does not give you legal ownership of a company. Instead, you are buying a digital asset you are betting will rise in value. Some cryptocurrencies may also offer some practical utility, like the ability to exchange them for goods and services. Company Filings Buy, sell & manage your Bitcoin and more crypto assets with one single app – Ledger Live. Later this year, users will be able to pay using cryptocurrencies in the PicPay app, and will be able to complete crypto transfers. Copyright © 2023 FCA. All rights reserved. Investing in cryptocurrencies involves purchasing and holding digital assets with the expectation that their value will increase over time. This long-term strategy banks on the belief that as cryptocurrencies become more widely adopted and integrated into technological and financial systems, their demand and, consequently, their value will rise. Investors aim to profit by eventually selling their holdings at a higher price than the cost at which they were acquired, realizing a capital gain. This approach requires patience, a strong belief in the underlying technology or utility of the asset, and a tolerance for the market’s inherent volatility.
I am no longer certain where you’re getting your info, however great topic.
I needs to spend a while finding out much more or working out more.
Thank you for wonderful information I was on the
lookout for this information for my mission.
https://canadadrugs.pro/# list of safe online pharmacies
cheap prednisolone 5mg buy generic prednisolone for sale cheap prednisolone online
cheap ed drugs: cheap erectile dysfunction pill – medicine for erectile
http://edpill.cheap/# best over the counter ed pills
india online pharmacy indian pharmacy reputable indian online pharmacy
http://edpill.cheap/# cheap erectile dysfunction pills
mexican mail order pharmacies: mexico drug stores pharmacies – mexican online pharmacies prescription drugs
cost clomid 100mg cost clomid clomiphene 50mg cheap
online canadian pharmacy canadian pharmacies online canadian pharmacy online
https://medicinefromindia.store/# online pharmacy india
Good info. Lucky me I reach on your website by accident, I bookmarked it.
buy prescription drugs from india п»їlegitimate online pharmacies india Online medicine home delivery
canada rx pharmacy world: canadian mail order pharmacy – canadian pharmacy near me
https://canadianinternationalpharmacy.pro/# canadian pharmacy com
viagra without doctor prescription amazon: generic cialis without a doctor prescription – prescription drugs
http://medicinefromindia.store/# india pharmacy
http://medicinefromindia.store/# best online pharmacy india
https://canadianinternationalpharmacy.pro/# ed drugs online from canada
mexican pharmacy without prescription non prescription ed pills viagra without doctor prescription
https://medicinefromindia.store/# indian pharmacies safe
indianpharmacy com: Online medicine order – india pharmacy mail order
how to get prescription drugs without doctor generic cialis without a doctor prescription buy prescription drugs online legally
how to get prescription drugs without doctor: ed prescription drugs – viagra without a doctor prescription
http://canadianinternationalpharmacy.pro/# canadian pharmacy meds
gabapentin 800mg pills neurontin 100mg usa gabapentin online
mexican border pharmacies shipping to usa mexican pharmacy mexican mail order pharmacies
gnc ed pills: ed medications online – ed meds online
http://medicinefromindia.store/# reputable indian pharmacies
http://edwithoutdoctorprescription.pro/# discount prescription drugs
the canadian pharmacy canadian pharmacy review recommended canadian pharmacies
http://medicinefromindia.store/# Online medicine home delivery
top erection pills: best ed drugs – ed treatment pills
canadian family pharmacy adderall canadian pharmacy canadian valley pharmacy
Your blog is a constant source of inspiration and knowledge. Thank you!
https://medicinefromindia.store/# indian pharmacy paypal
pills for ed erection pills viagra online cure ed
viagra 50mg cost viagra australia sildenafil 50mg cost
http://medicinefromindia.store/# indian pharmacy
prescription without a doctor’s prescription generic cialis without a doctor prescription prescription drugs without doctor approval
http://canadianinternationalpharmacy.pro/# canadian online pharmacy
generic viagra without a doctor prescription: cheap cialis – buy cheap prescription drugs online
https://medicinefromindia.store/# indian pharmacy
mail order pharmacy india п»їlegitimate online pharmacies india world pharmacy india
buy lasix paypal lasix 40mg cost cost furosemide 100mg
http://edpill.cheap/# men’s ed pills
mexican mail order pharmacies mexican pharmaceuticals online п»їbest mexican online pharmacies
https://canadianinternationalpharmacy.pro/# canadian pharmacy online
best ed pills at gnc non prescription ed drugs otc ed pills
canadian drug: canadian online drugstore – canadianpharmacymeds
https://edpill.cheap/# cheapest ed pills online
п»їbest mexican online pharmacies buying prescription drugs in mexico reputable mexican pharmacies online
http://edwithoutdoctorprescription.pro/# ed meds online without doctor prescription
https://medicinefromindia.store/# world pharmacy india
buy semaglutide 14mg pills semaglutide 14mg oral order semaglutide 14mg online cheap
canada drug pharmacy prescription drugs canada buy online canadian mail order pharmacy
http://edpill.cheap/# ed medication online
world pharmacy india buy prescription drugs from india buy prescription drugs from india
canadian pharmacy ltd: reliable canadian pharmacy reviews – best canadian pharmacy
https://medicinefromindia.store/# india online pharmacy
order generic acticlate order generic doxycycline 100mg buy vibra-tabs pills
http://edpill.cheap/# ed meds online
ed meds online without prescription or membership ed pills without doctor prescription prescription drugs
pharmacies in mexico that ship to usa buying prescription drugs in mexico reputable mexican pharmacies online
mexican drugstore online mexican pharmaceuticals online medicine in mexico pharmacies
https://mexicanph.com/# mexico pharmacies prescription drugs
reputable mexican pharmacies online
mexico pharmacies prescription drugs mexican drugstore online purple pharmacy mexico price list
mexican pharmacy mexico pharmacies prescription drugs medicine in mexico pharmacies
mexico drug stores pharmacies best mexican online pharmacies mexico pharmacies prescription drugs
I’m amazed by the depth and breadth of your knowledge. Thanks for sharing!
buying prescription drugs in mexico online mexican pharmacy mexican online pharmacies prescription drugs
purple pharmacy mexico price list buying prescription drugs in mexico mexican mail order pharmacies
order vardenafil generic order vardenafil for sale order vardenafil online cheap
I was suggested this website by my cousin. I am not certain whether this
submit is written via him as no one else realize such designated approximately my trouble.
You are incredible! Thank you!
http://mexicanph.shop/# buying prescription drugs in mexico
mexican mail order pharmacies
mexico drug stores pharmacies mexico pharmacy mexican pharmaceuticals online
pharmacies in mexico that ship to usa buying prescription drugs in mexico online mexico drug stores pharmacies
slots casino games spins real money online casino free spins no deposit
buying from online mexican pharmacy buying prescription drugs in mexico medicine in mexico pharmacies
pharmacies in mexico that ship to usa mexican drugstore online purple pharmacy mexico price list
This is one of the most comprehensive articles I’ve read on this topic. Kudos!
mexican border pharmacies shipping to usa mexico drug stores pharmacies п»їbest mexican online pharmacies
http://mexicanph.com/# medication from mexico pharmacy
reputable mexican pharmacies online
mexico drug stores pharmacies mexican pharmaceuticals online mexican border pharmacies shipping to usa
mexican border pharmacies shipping to usa mexico drug stores pharmacies best online pharmacies in mexico
reputable mexican pharmacies online mexico drug stores pharmacies buying prescription drugs in mexico
buying prescription drugs in mexico online medication from mexico pharmacy reputable mexican pharmacies online
order plaquenil 200mg sale purchase hydroxychloroquine online cheap buy hydroxychloroquine generic
https://mexicanph.com/# best online pharmacies in mexico
medication from mexico pharmacy
mexican mail order pharmacies medicine in mexico pharmacies mexican pharmaceuticals online
reputable mexican pharmacies online purple pharmacy mexico price list mexican mail order pharmacies
generic lyrica buy lyrica paypal pregabalin 75mg generic
п»їbest mexican online pharmacies mexico drug stores pharmacies medicine in mexico pharmacies
medication from mexico pharmacy reputable mexican pharmacies online buying prescription drugs in mexico
pharmacies in mexico that ship to usa buying prescription drugs in mexico online pharmacies in mexico that ship to usa
mexico pharmacies prescription drugs mexican rx online purple pharmacy mexico price list
reputable mexican pharmacies online mexican pharmaceuticals online purple pharmacy mexico price list
http://mexicanph.shop/# mexican online pharmacies prescription drugs
pharmacies in mexico that ship to usa
mexico drug stores pharmacies mexico pharmacy buying prescription drugs in mexico
best online pharmacies in mexico mexico pharmacies prescription drugs medication from mexico pharmacy
purple pharmacy mexico price list mexican online pharmacies prescription drugs mexican pharmaceuticals online
mexican mail order pharmacies mexico drug stores pharmacies best online pharmacies in mexico
purple pharmacy mexico price list mexico drug stores pharmacies purple pharmacy mexico price list
mexico pharmacy best online pharmacies in mexico mexican pharmacy
medicine in mexico pharmacies mexico pharmacies prescription drugs mexico pharmacies prescription drugs
mexican border pharmacies shipping to usa mexico pharmacies prescription drugs mexican online pharmacies prescription drugs
mexico pharmacy buying prescription drugs in mexico mexico pharmacy
cost cialis 20mg order cialis 10mg without prescription buy cialis canada
buying prescription drugs in mexico best mexican online pharmacies medicine in mexico pharmacies
mexico pharmacy mexican pharmacy buying prescription drugs in mexico online
triamcinolone us aristocort 4mg drug oral triamcinolone
http://mexicanph.shop/# buying prescription drugs in mexico
reputable mexican pharmacies online
mexican rx online mexican online pharmacies prescription drugs mexico pharmacy
buying from online mexican pharmacy buying prescription drugs in mexico online mexican mail order pharmacies
mexican online pharmacies prescription drugs mexican border pharmacies shipping to usa mexican rx online
buying prescription drugs in mexico online buying prescription drugs in mexico online mexico pharmacy
buying prescription drugs in mexico mexican pharmaceuticals online buying prescription drugs in mexico online
mexico pharmacy mexican drugstore online medication from mexico pharmacy
buying prescription drugs in mexico mexican pharmacy best online pharmacies in mexico
mexican drugstore online buying from online mexican pharmacy mexican mail order pharmacies
medication from mexico pharmacy mexican online pharmacies prescription drugs mexico drug stores pharmacies
https://mexicanph.shop/# pharmacies in mexico that ship to usa
mexican drugstore online
reputable mexican pharmacies online buying from online mexican pharmacy mexican border pharmacies shipping to usa
mexican pharmaceuticals online mexico pharmacies prescription drugs medicine in mexico pharmacies
mexican border pharmacies shipping to usa pharmacies in mexico that ship to usa mexican online pharmacies prescription drugs
pharmacies in mexico that ship to usa mexican rx online medicine in mexico pharmacies
medicine in mexico pharmacies mexican rx online mexican pharmaceuticals online
pharmacies in mexico that ship to usa mexico drug stores pharmacies pharmacies in mexico that ship to usa
reputable mexican pharmacies online medicine in mexico pharmacies pharmacies in mexico that ship to usa
purple pharmacy mexico price list mexican border pharmacies shipping to usa purple pharmacy mexico price list
cheap cenforce 100mg buy cenforce cheap purchase cenforce without prescription
mexican pharmacy mexican border pharmacies shipping to usa mexican rx online
best online pharmacies in mexico best online pharmacies in mexico buying prescription drugs in mexico
how to buy desloratadine desloratadine cheap buy clarinex 5mg pill
mexican rx online reputable mexican pharmacies online buying from online mexican pharmacy
buying from online mexican pharmacy mexican online pharmacies prescription drugs mexican drugstore online
mexico drug stores pharmacies mexican drugstore online buying prescription drugs in mexico
reputable mexican pharmacies online mexican mail order pharmacies mexico drug stores pharmacies
https://mexicanph.shop/# mexico drug stores pharmacies
mexican pharmaceuticals online
mexican border pharmacies shipping to usa mexican pharmaceuticals online mexico drug stores pharmacies
mexican mail order pharmacies mexico drug stores pharmacies purple pharmacy mexico price list
buying prescription drugs in mexico online mexican drugstore online reputable mexican pharmacies online
buying prescription drugs in mexico online medicine in mexico pharmacies mexican pharmaceuticals online
mexico drug stores pharmacies buying from online mexican pharmacy mexico drug stores pharmacies
medication from mexico pharmacy medication from mexico pharmacy reputable mexican pharmacies online
pharmacies in mexico that ship to usa best online pharmacies in mexico mexican rx online
mexico pharmacy medication from mexico pharmacy best mexican online pharmacies
purchase chloroquine online brand chloroquine chloroquine pills
mexican rx online buying prescription drugs in mexico online buying from online mexican pharmacy
reputable mexican pharmacies online mexican online pharmacies prescription drugs medication from mexico pharmacy
mexican pharmaceuticals online mexican pharmacy buying from online mexican pharmacy
mexican pharmacy reputable mexican pharmacies online mexican drugstore online
mexican pharmaceuticals online mexican pharmaceuticals online buying prescription drugs in mexico
order generic claritin 10mg buy loratadine sale loratadine online buy
http://amoxil.cheap/# amoxicillin capsules 250mg
buy amoxicillin online uk: generic amoxicillin over the counter – amoxicillin tablet 500mg
http://amoxil.cheap/# amoxicillin script
buy amoxicillin canada: where can i get amoxicillin 500 mg – amoxicillin medicine
order amoxicillin online no prescription medicine amoxicillin 500 amoxicillin 500 mg online
https://furosemide.guru/# lasix furosemide 40 mg
prednisone 30 mg tablet: prednisone cream brand name – buy prednisone online fast shipping
https://furosemide.guru/# furosemide 100mg
amoxicillin for sale amoxicillin 500 coupon 875 mg amoxicillin cost
lasix furosemide: lasix side effects – furosemide 100 mg
stromectol tab price: oral ivermectin cost – stromectol price
http://amoxil.cheap/# amoxicillin 200 mg tablet
https://lisinopril.top/# lisinopril 30 mg daily
https://furosemide.guru/# furosemida 40 mg
furosemide 100 mg Buy Lasix generic lasix
lisinopril 20mg pill: lisinopril for sale uk – cost of lisinopril 2.5 mg
order glycomet 500mg generic metformin 500mg canada purchase glycomet
https://furosemide.guru/# furosemida 40 mg
ivermectin lotion for scabies: ivermectin 8000 – ivermectin where to buy
lisinopril generic drug: generic lisinopril 3973 – lisinopril 2.5 mg price
amoxicillin discount coupon amoxicillin medicine over the counter amoxicillin cost australia
order dapoxetine 30mg without prescription cost misoprostol how to get misoprostol without a prescription
https://lisinopril.top/# buy lisinopril 20 mg
prednisone in canada: prednisone 475 – prednisone uk buy
https://furosemide.guru/# generic lasix
http://stromectol.fun/# ivermectin 9 mg tablet
amoxicillin without rx: amoxicillin buy online canada – amoxicillin 500 coupon
lisinopril sale lisinopril 50 mg price zestoretic
lasix 100 mg: Over The Counter Lasix – lasix uses
http://lisinopril.top/# lisinopril online
I read this piece of writing completely on the topic of the difference of hottest and
earlier technologies, it’s amazing article.
https://lisinopril.top/# buy lisinopril online no prescription
order lisinopril online united states: lisinopril in usa – lisinopril price 10 mg
buy lisinopril without a prescription: cost for 20 mg lisinopril – lisinopril 20 mg prices
amoxicillin 500mg amoxicillin online canada purchase amoxicillin online
http://amoxil.cheap/# how much is amoxicillin prescription
no prescription prednisone canadian pharmacy: 5mg prednisone – prednisone 5mg price
https://buyprednisone.store/# no prescription online prednisone
lisinopril 80mg tablet zestril cost price price of lisinopril 20 mg
http://furosemide.guru/# lasix tablet
amoxicillin 800 mg price: prescription for amoxicillin – amoxicillin 500 mg price
lisinopril 3972: lisinopril 40 mg canada – compare zestril prices
http://buyprednisone.store/# mail order prednisone
purchase orlistat online cheap generic orlistat 60mg buy generic diltiazem online
atorvastatin 40mg for sale lipitor 40mg price order generic lipitor 10mg
buy amoxicillin 500mg online: how much is amoxicillin – amoxicillin 500 mg for sale
https://furosemide.guru/# buy furosemide online
can i purchase amoxicillin online where can i buy amoxocillin rexall pharmacy amoxicillin 500mg
zestril 20 mg tablet: lisinopril metoprolol – lisinopril price comparison
http://amoxil.cheap/# 875 mg amoxicillin cost
where to buy ivermectin: ivermectin 3mg tablets – stromectol 3mg tablets
http://buyprednisone.store/# prednisone uk over the counter
where to buy stromectol buy oral ivermectin stromectol 3 mg price
lisinopril 20 mg generic: lisinopril capsule – zestril 25 mg
http://buyprednisone.store/# prednisone 12 mg
https://amoxil.cheap/# amoxicillin 500mg price in canada
buy lisinopril online uk: 60 lisinopril cost – buy lisinopril online canada
https://amoxil.cheap/# amoxicillin 500mg prescription
prednisone 10 mg prednisone 20 mg without prescription buy prednisone online without a prescription
cost amlodipine norvasc ca order norvasc 10mg generic
https://buyprednisone.store/# buy prednisone online australia
lisinopril without an rx: cost of lisinopril in mexico – cost of brand name lisinopril
https://stromectol.fun/# ivermectin price
generic amoxicillin online: amoxicillin 500mg capsules – amoxicillin 250 mg capsule
lisinopril 5 mg over the counter lisinopril 10 lisinopril 3.5 mg
https://amoxil.cheap/# cost of amoxicillin
http://buyprednisone.store/# prednisone in mexico
amoxicillin 800 mg price: generic amoxicillin cost – amoxicillin 500mg over the counter
https://lisinopril.top/# lisinopril 10 mg prices
lisinopril 10mg online: zestril price – zestril 20 mg price canadian pharmacy
where can i buy amoxicillin over the counter amoxicillin 250 mg capsule amoxicillin without a doctors prescription
https://buyprednisone.store/# 10mg prednisone daily
ivermectin 0.08%: ivermectin 3 – stromectol cost
brand zovirax 800mg zyloprim 100mg brand zyloprim price
https://amoxil.cheap/# amoxicillin script
ivermectin for humans: generic stromectol – stromectol ivermectin
over the counter prednisone cheap: prednisone cream brand name – prednisone in mexico
generic ivermectin ivermectin 6 ivermectin 18mg
http://furosemide.guru/# lasix 100 mg
buy zestril 5mg generic order generic lisinopril buy lisinopril 2.5mg without prescription
lisinopril 25 mg: lisinopril 20mg for sale – can i buy generic lisinopril online
https://stromectol.fun/# price of ivermectin liquid
Your blog consistently provides valuable insights. We’re thrilled to be avid readers from Asheville!
can you buy amoxicillin over the counter in canada amoxicillin buy no prescription medicine amoxicillin 500mg
zestril 10 mg: lisinopril 10 mg without prescription – zestril discount
ivermectin 18mg: stromectol buy – stromectol where to buy
http://stromectol.fun/# ivermectin lice oral
lisinopril prescription coupon: prinivil cost – lisinopril no prescription
lasix medication: Buy Furosemide – lasix 100 mg tablet
I just wanted to take a moment to express my appreciation for your fantastic blog. Asheville can’t get enough!
http://buyprednisone.store/# prednisone 2 mg daily
order crestor 10mg online rosuvastatin 10mg generic ezetimibe online
buy amoxicillin online cheap: amoxicillin 1000 mg capsule – buy amoxicillin online uk
http://stromectol.fun/# stromectol 3mg
http://amoxil.cheap/# buying amoxicillin in mexico
where to buy amoxicillin 500mg buy amoxicillin online no prescription amoxicillin 500 mg price
order prednisone from canada: where can i buy prednisone without prescription – where to get prednisone
generic prilosec 20mg order prilosec 10mg sale order omeprazole 20mg pill
http://indianph.com/# top 10 pharmacies in india
india pharmacy
motilium 10mg tablet brand tetracycline 500mg brand sumycin 500mg
pharmacy website india world pharmacy india cheapest online pharmacy india
indian pharmacy online п»їlegitimate online pharmacies india buy prescription drugs from india
http://indianph.xyz/# world pharmacy india
world pharmacy india
top online pharmacy india pharmacy website india indian pharmacies safe
http://indianph.com/# online shopping pharmacy india
online shopping pharmacy india
https://indianph.xyz/# top online pharmacy india
top 10 online pharmacy in india
lopressor 100mg brand buy lopressor cheap metoprolol 50mg usa
https://indianph.com/# cheapest online pharmacy india
top online pharmacy india
best online pharmacy india buy medicines online in india pharmacy website india
Thank you for consistently producing such high-quality content.
https://indianph.com/# top 10 pharmacies in india
indian pharmacy paypal
https://indianph.xyz/# Online medicine home delivery
indian pharmacies safe
buy medicines online in india india pharmacy mail order best india pharmacy
india online pharmacy indian pharmacy online indian pharmacies safe
buy cheap flexeril buy cyclobenzaprine online baclofen usa
https://nolvadex.guru/# tamoxifen citrate
atenolol order online atenolol 50mg brand buy tenormin 50mg pill
http://nolvadex.guru/# raloxifene vs tamoxifen
diflucan cost canada: buying diflucan over the counter – diflucan 150mg tab
nolvadex price benefits of tamoxifen nolvadex vs clomid
https://nolvadex.guru/# does tamoxifen cause joint pain
tamoxifen cyp2d6: who should take tamoxifen – tamoxifen dose
https://doxycycline.auction/# generic doxycycline
diflucan from india diflucan tablets australia diflucan price in india
https://diflucan.pro/# diflucan 100mg
http://cipro.guru/# cipro online no prescription in the usa
buy cytotec in usa buy cytotec in usa buy cytotec in usa
https://cytotec24.com/# buy cytotec in usa
buy cytotec online fast delivery: buy misoprostol over the counter – Misoprostol 200 mg buy online
https://doxycycline.auction/# doxycycline order online
https://doxycycline.auction/# where can i get doxycycline
buy generic ciprofloxacin cipro ciprofloxacin cipro pharmacy
https://nolvadex.guru/# tamoxifen therapy
order doxycycline online: where to get doxycycline – doxycycline 100mg price
http://cytotec24.shop/# buy cytotec online
doxycycline generic vibramycin 100 mg doxycycline generic
http://cytotec24.com/# purchase cytotec
medrol cost in usa medrol 8mg over counter order depo-medrol pill
https://diflucan.pro/# where to buy diflucan
http://cytotec24.shop/# buy misoprostol over the counter
Your blog is a constant source of inspiration and knowledge. Thank you!
can i buy diflucan over the counter uk diflucan 100 mg tablet diflucan.com
I learned a lot, and now I’m curious about what else you could teach me. Your intelligence is as captivating as your prose.
http://doxycycline.auction/# doxycycline
toradol for sale order toradol without prescription buy gloperba cheap
https://doxycycline.auction/# how to buy doxycycline online
https://cytotec24.shop/# buy cytotec pills
cipro ciprofloxacin ciprofloxacin generic cipro pharmacy
http://doxycycline.auction/# doxycycline 100mg online
http://doxycycline.auction/# doxycycline hyc 100mg
where can you get diflucan where can you get diflucan over the counter diflucan rx online
https://lanarhoades.fun/# lana rhoades filmleri
https://abelladanger.online/# Abella Danger
Angela White izle: abella danger video – abella danger izle
http://lanarhoades.fun/# lana rhoades filmleri
https://abelladanger.online/# abella danger izle
Angela White video: abella danger filmleri – abella danger izle
essay writers online letter editing help with term paper
http://angelawhite.pro/# Angela White video
http://sweetiefox.online/# Sweetie Fox
https://abelladanger.online/# abella danger izle
lana rhoades: lana rhoades izle – lana rhoades filmleri
https://sweetiefox.online/# Sweetie Fox izle
https://abelladanger.online/# abella danger video
http://sweetiefox.online/# Sweetie Fox izle
lana rhoades video: lana rhoades izle – lana rhoades
https://abelladanger.online/# abella danger video
inderal 10mg oral clopidogrel 75mg usa buy clopidogrel tablets
https://angelawhite.pro/# Angela White filmleri
https://angelawhite.pro/# Angela White
Angela Beyaz modeli: ?????? ???? – Angela White izle
https://abelladanger.online/# Abella Danger
http://sweetiefox.online/# sweeti fox
lana rhoades: lana rhodes – lana rhodes
http://evaelfie.pro/# eva elfie
http://sweetiefox.online/# Sweetie Fox modeli
http://sweetiefox.online/# Sweetie Fox izle
http://abelladanger.online/# abella danger izle
Angela White izle: Angela White filmleri – ?????? ????
http://abelladanger.online/# abella danger filmleri
http://sweetiefox.online/# Sweetie Fox izle
http://angelawhite.pro/# Angela White
Angela White: Abella Danger – abella danger izle
https://evaelfie.pro/# eva elfie modeli
http://abelladanger.online/# Abella Danger
order methotrexate 10mg for sale coumadin buy online buy generic coumadin online
?????? ????: Angela White video – Angela White filmleri
http://angelawhite.pro/# Angela White
buy generic mobic celebrex ca brand celebrex 200mg
https://abelladanger.online/# abella danger filmleri
Your insights light up my intellect like fireworks. Thanks for the show!
eva elfie modeli: eva elfie modeli – eva elfie
http://angelawhite.pro/# Angela White filmleri
https://abelladanger.online/# abella danger izle
https://sweetiefox.online/# swetie fox
https://evaelfie.pro/# eva elfie modeli
http://abelladanger.online/# abella danger izle
Your insights are like keys, unlocking new perspectives and ideas I hadn’t considered.
https://lanarhoades.fun/# lana rhoades
http://lanarhoades.fun/# lana rhoades filmleri
http://evaelfie.pro/# eva elfie modeli
Angela Beyaz modeli: abella danger izle – abella danger video
Your insights are like a sunrise, bringing light and warmth to new ideas.
metoclopramide 10mg us maxolon uk losartan 25mg cheap
Your blog is like a favorite coffee shop where the drinks are always warm and the atmosphere is inviting.
https://angelawhite.pro/# Angela Beyaz modeli
lana rhoades izle: lana rhoades – lana rhoades video
https://abelladanger.online/# abella danger izle
mia malkova videos: mia malkova girl – mia malkova videos
mia malkova new video: mia malkova latest – mia malkova videos
flomax 0.2mg cost flomax 0.4mg pills order celebrex 200mg generic
https://sweetiefox.pro/# sweetie fox video
lana rhoades boyfriend: lana rhoades unleashed – lana rhoades hot
http://miamalkova.life/# mia malkova latest
our time dating website: https://miamalkova.life/# mia malkova
eva elfie hot: eva elfie – eva elfie hd
Hi there, i read your blog occasionally and i own a similar one and i was just wondering if you get a lot of spam responses? If so how do you stop it, any plugin or anything you can advise? I get so much lately it’s driving me crazy so any assistance is very much appreciated.
sweetie fox: sweetie fox – ph sweetie fox
https://sweetiefox.pro/# ph sweetie fox
sweetie fox video: fox sweetie – sweetie fox
This article was a delightful enjoy reading. Your passion is clearly visible!
Making complex concepts readable is no small feat. It’s like you know exactly how to tickle my brain.
christian dating sites: http://miamalkova.life/# mia malkova new video
https://miamalkova.life/# mia malkova videos
eva elfie: eva elfie photo – eva elfie hd
mia malkova only fans: mia malkova full video – mia malkova movie
sweetie fox cosplay: sweetie fox full video – fox sweetie
https://evaelfie.site/# eva elfie hd
free chat sites dating: http://evaelfie.site/# eva elfie full videos
mia malkova girl: mia malkova – mia malkova full video
lana rhoades full video: lana rhoades solo – lana rhoades hot
http://evaelfie.site/# eva elfie full video
Your writing style is captivating! I was engaged from start to finish.
eva elfie: eva elfie hd – eva elfie full video
You tackled a complex issue with elegance and insight. I feel much more informed after reading your post.
Your post provoked a lot of thought and taught me something new. Thank you for such engaging content.
purchase nexium buy topiramate 200mg generic buy topamax generic
Your creativity shines through, making me wonder what else you could do with such a vivid imagination.
https://lanarhoades.pro/# lana rhoades pics
dating web: http://miamalkova.life/# mia malkova latest
cheap zofran zofran ca aldactone 100mg sale
sweetie fox full: sweetie fox full – sweetie fox full
sweetie fox full: sweetie fox – sweetie fox new
http://lanarhoades.pro/# lana rhoades videos
eva elfie videos: eva elfie full video – eva elfie
date me site: https://lanarhoades.pro/# lana rhoades unleashed
http://lanarhoades.pro/# lana rhoades pics
lana rhoades unleashed: lana rhoades solo – lana rhoades unleashed
order generic imitrex 25mg buy levaquin 500mg generic levaquin 250mg tablet
mia malkova new video: mia malkova movie – mia malkova movie
eva elfie: eva elfie full video – eva elfie hot
how to get simvastatin without a prescription how to buy valacyclovir brand valacyclovir 1000mg
https://lanarhoades.pro/# lana rhoades full video
local dating site: https://miamalkova.life/# mia malkova photos
lana rhoades: lana rhoades boyfriend – lana rhoades
https://miamalkova.life/# mia malkova only fans
mia malkova latest: mia malkova photos – mia malkova full video
https://aviatormocambique.site/# aviator mz
jogo de aposta: jogos que dão dinheiro – jogos que dão dinheiro
http://aviatorjogar.online/# aviator jogo
generic avodart buy dutasteride sale buy ranitidine 300mg generic
site de apostas: melhor jogo de aposta – aplicativo de aposta
http://aviatormocambique.site/# aviator mocambique
propecia 5mg uk where can i buy diflucan order diflucan 200mg generic
Testament to your expertise and hard work, or your ability to make me feel utterly unaccomplished.
aviator: aviator online – aviator
Neon Flying Squid
http://www.saghyendre.hu/en/2019/11/10/szentendre-2/
https://pinupcassino.pro/# pin-up casino entrar
https://jogodeaposta.fun/# jogo de aposta
pin-up cassino: pin up bet – aviator oficial pin up
Every word you write sparkles with insight, like stars in my night sky. Can’t wait to navigate more skies together.
aviator oficial pin up: aviator oficial pin up – pin up cassino online
aplicativo de aposta: jogo de aposta – deposito minimo 1 real
https://aviatorghana.pro/# aviator sportybet ghana
pin up aviator: pin-up cassino – aviator pin up casino
https://aviatorghana.pro/# aviator bet
I’m genuinely impressed by the depth of your analysis. Great work!
buy acillin buy penicillin for sale amoxil online order
aviator sportybet ghana: aviator – aviator game
The passion you pour into your posts is like a flame, igniting curiosity and warming the soul.
aviator bet malawi: aviator malawi – aviator betting game
aviator bet: aviator game – estrela bet aviator
jogar aviator online: aviator pin up – estrela bet aviator
pin up aviator: jogar aviator Brasil – pin up aviator
aviator bet malawi: aviator betting game – aviator
estrela bet aviator: aviator bet – pin up aviator
http://aviatorjogar.online/# aviator bet
melhor jogo de aposta: aplicativo de aposta – jogos que dao dinheiro
You have a gift for explaining things in an understandable way. Thank you!
brand baycip – sulfamethoxazole usa purchase amoxiclav pill
aviator bet: aviator game online – aviator bet
generic zithromax 500mg india: zithromax 500 mg for sale – zithromax 500 mg lowest price drugstore online
aviator pin up casino: pin up – pin-up casino
https://aviatormocambique.site/# como jogar aviator
zithromax 250 price: zithromax allergic reaction zithromax z-pak price without insurance
pin up cassino online: pin-up casino – pin-up
aviator game bet: aviator betting game – aviator login
https://pinupcassino.pro/# pin up aviator
generic zithromax online paypal: zithromax and pregnancy zithromax buy online
baycip oral – purchase septra for sale buy augmentin generic
This article was a delightful enjoy reading. Your passion is clearly visible!
aviator pin up: aviator betano – aviator jogar
aviator: aviator online – como jogar aviator em moçambique
Your work is truly inspirational. I appreciate the depth you bring to your topics.
top 10 pharmacies in india: india pharmacy – online shopping pharmacy india indianpharm.store
Unique perspective? Check. Making me rethink my life choices? Double-check.
canadian online pharmacy: Canada pharmacy – canadian pharmacy ratings canadianpharm.store
Completely aligned with the opinions above – this post is a true gem!
mexican drugstore online: mexican drugstore online – п»їbest mexican online pharmacies mexicanpharm.shop
The hard work you put into this post is as admirable as your commitment to quality. It’s very attractive.
top 10 pharmacies in india: Online India pharmacy – mail order pharmacy india indianpharm.store
http://mexicanpharm24.com/# mexico pharmacy mexicanpharm.shop
http://indianpharm24.shop/# п»їlegitimate online pharmacies india indianpharm.store
A breath of fresh air, or what I needed after being suffocated by mediocrity.
https://indianpharm24.com/# online pharmacy india indianpharm.store
medication from mexico pharmacy: order online from a Mexican pharmacy – п»їbest mexican online pharmacies mexicanpharm.shop
legitimate canadian mail order pharmacy: My Canadian pharmacy – canada pharmacy reviews canadianpharm.store
https://mexicanpharm24.com/# mexican mail order pharmacies mexicanpharm.shop
http://mexicanpharm24.shop/# mexico drug stores pharmacies mexicanpharm.shop
https://indianpharm24.com/# world pharmacy india indianpharm.store
order metronidazole 200mg generic – buy amoxicillin online cheap zithromax usa
online shopping pharmacy india: Online medicine home delivery – online shopping pharmacy india indianpharm.store
Thank you for consistently producing such high-quality content.
best canadian online pharmacy reviews Canadian pharmacy prices canada drugs online review canadianpharm.store
http://mexicanpharm24.shop/# п»їbest mexican online pharmacies mexicanpharm.shop
http://canadianpharmlk.shop/# canadian pharmacy victoza canadianpharm.store
http://indianpharm24.shop/# indianpharmacy com indianpharm.store
Thank you for shedding light on this subject. Your perspective is refreshing!
https://canadianpharmlk.com/# safe reliable canadian pharmacy canadianpharm.store
Your passion for this subject shines through your words. Inspiring!
best canadian pharmacy online: Canada pharmacy – pharmacy canadian superstore canadianpharm.store
canada rx pharmacy: My Canadian pharmacy – canadian pharmacy review canadianpharm.store
http://canadianpharmlk.com/# my canadian pharmacy reviews canadianpharm.store
https://canadianpharmlk.shop/# legit canadian pharmacy canadianpharm.store
http://indianpharm24.com/# india pharmacy indianpharm.store
https://mexicanpharm24.shop/# medicine in mexico pharmacies mexicanpharm.shop
canadian valley pharmacy: List of Canadian pharmacies – canadian drugs online canadianpharm.store
http://indianpharm24.shop/# top 10 pharmacies in india indianpharm.store
https://mexicanpharm24.shop/# mexican border pharmacies shipping to usa mexicanpharm.shop
canadian pharmacy no scripts canadian pharmacy canadian pharmacy 365 canadianpharm.store
I’m always excited to see your posts in my feed. Another excellent article!
canadian pharmacy store: ed meds online canada – canadian pharmacy 24h com canadianpharm.store
https://canadianpharmlk.shop/# canadianpharmacy com canadianpharm.store
https://canadianpharmlk.com/# best canadian pharmacy online canadianpharm.store
https://indianpharm24.com/# top 10 online pharmacy in india indianpharm.store
http://mexicanpharm24.com/# pharmacies in mexico that ship to usa mexicanpharm.shop
brand ciprofloxacin – doryx uk erythromycin brand
can i buy amoxicillin over the counter in australia: amoxicillin for dogs – 875 mg amoxicillin cost
amoxicillin 800 mg price: amoxicillin tablet 500mg – over the counter amoxicillin
https://clomidst.pro/# can i buy clomid without prescription
mail order prednisone: 1 mg prednisone daily – can i buy prednisone online without a prescription
buy prednisone 10 mg: side effects prednisone – prednisone for sale without a prescription
https://prednisonest.pro/# prednisone 2 mg daily
prednisolone prednisone: does prednisone make you tired – order prednisone from canada
buy amoxicillin over the counter uk amoxicillin 500 coupon ampicillin amoxicillin
buy amoxicillin online with paypal: amoxicillin cost australia – amoxicillin online canada
amoxicillin script: ampicillin vs amoxicillin – can i buy amoxicillin online
buy prednisone online canada: side effect of prednisone – prednisone 475
This article is a perfect blend of informative and entertaining. Well done!
2.5 mg prednisone daily: prednisone 20 mg dosage instructions – average cost of prednisone 20 mg
http://amoxilst.pro/# how to get amoxicillin over the counter
buy valacyclovir 500mg generic – nemasole ca buy acyclovir pill
where to buy prednisone in canada: alcohol and prednisone – prednisone for sale in canada
http://clomidst.pro/# can you buy clomid now
prednisone tabs 20 mg: prednisone 300mg – prednisone 21 pack
where can you buy amoxicillin over the counter: amoxicillin used for – generic amoxicillin
where can i buy cheap clomid without prescription: cost cheap clomid without insurance – can i buy cheap clomid tablets
price of prednisone 5mg prednisone 5084 prednisone tablets india
how much is prednisone 5mg: what is prednisone – ordering prednisone
http://prednisonest.pro/# 100 mg prednisone daily
prednisone 4mg: buy prednisone online usa – prednisone 20mg prices
Neon Flying Squid
https://durainformativa.com/el-poder-ejecutivo-presentara-un-presupuesto-general-por-un-monto-de-billon-46-mil-millones-inferior-al-actual/
amoxicillin 500 mg tablets: amoxicillin 500mg price – amoxicillin over counter
buy clomid without prescription: where to get generic clomid without prescription – cost cheap clomid
where can i buy clomid: long-term use of clomid in males – cost of clomid prices
http://clomidst.pro/# order generic clomid pill
where can i get clomid without prescription: clomid symptoms – can i order clomid tablets
Twój Ford został zaprojektowany z najwyższą precyzją, dzięki czemu możesz dostosować go własnego stylu życia i aktywności. Spraw, aby Twój samochód był unikalny – dokładnie tak, jak Ty. Uzupełnij wyposażenie w oryginalne części i akcesoria marki Ford w Auto Brzezińska. Dysponujemy szerokim wyborem produktów – od praktycznych, stylowych dodatków niezbędnych na co dzień, po eleganckie i wygodne rozwiązania i najnowsze akcesoria – zarówno wewnątrz, jak i na zewnątrz Twojego pojazdu. Inter Pack New Spider 4 składana platforma rowerowa służąca do przewozu 4 rowerów.Platform.. W MH Motors możemy zamówić wszystkie poniższe akcesoria i dopasować je do Twojego modelu. Jeżeli chcesz poznać kompletną gamę produktów lub wybrać konkretną opcję, skontaktuj się z nami: oddzwonimy do Ciebie lub wyślemy e-mail.
https://www.lawyersclubindia.com/profile.asp?member_id=1029992
Al. Prymasa Tysiąclecia 66, Warszawa W tym miejscu możesz określić swoje preferencje w zakresie wykorzystywania przez nas plików cookies. Sklep AutoCentrum.online stworzona została z myślą o wygodzie klientów poszukujących samochodowych części zamiennych: nowych oryginalnych OEM, znacznie tańszych zamienników oraz elementów używanych. Wśród dostępnych produktów oferowanych przez nasz internetowy sklep motoryzacyjny znajdują się np. akumulatory, opony czy oleje silnikowe. Szeroka oferta dostępnych produktów sprawia, że praktycznie każdy klient znajdzie u nas podzespoły dostosowane do jego indywidualnych potrzeb oraz budżetu. 5600warsztatów na Świecie ZADZWOŃ DO NAS W naszym sklepie znajdziesz najróżniejsze używane części zamienne do samochodów. Oferujemy szeroki wybór produktów, które pasują do pojazdów różnych marek. Niezależnie od modelu i rocznika, u nas znajdziesz odpowiednie elementy, które pomogą utrzymać Twój pojazd w doskonałej kondycji.
prednisone brand name us: does prednisone cause constipation – 6 prednisone
where to get cheap clomid for sale: where can i buy generic clomid without insurance – can you get clomid no prescription
ivermectin 3 mg otc – order suprax where can i buy tetracycline
canadian pharmacy coupon Cheapest online pharmacy canadian pharmacy coupon code
best online ed medication: where can i get ed pills – online ed drugs
http://edpills.guru/# erectile dysfunction medicine online
http://pharmnoprescription.pro/# buying prescription medications online
erectile dysfunction pills for sale: ed online pharmacy – ed med online
online ed drugs: cheap boner pills – ed pills cheap
canadian pharmacy no prescription: canada pharmacy online – pharmacy discount coupons
https://pharmnoprescription.pro/# online pharmacy without prescription
buy prescription drugs without a prescription: no prescription pharmacy online – no prescription
get ed meds today: affordable ed medication – cheap ed pills online
https://pharmnoprescription.pro/# buy prescription drugs on line
cheap prescription drugs online: how to order prescription drugs from canada – online pharmacies no prescription
https://onlinepharmacy.cheap/# mail order prescription drugs from canada
ed medicines: buy erectile dysfunction pills – online ed treatments
oral metronidazole 400mg – azithromycin 500mg without prescription order azithromycin 250mg online
canadian pharmacy world coupons: canada pharmacy online – no prescription needed pharmacy
https://edpills.guru/# cheap boner pills
Neon Flying Squid
https://www.enbigi.com/tidak-perlu-obat-kuat-untuk-meningkatkan-vitalitas-pria-ini-rahasianya/
no prescription pharmacy paypal: canadian online pharmacy – canadian pharmacy no prescription
https://edpills.guru/# ed meds cheap
no prescription needed: canada drugs without prescription – no prescription medicine
pharmacy without prescription: online pharmacy delivery – canadian pharmacy discount coupon
Neon Flying Squid
http://www.liacibinong.com/program-kursus/?p=9036
https://onlinepharmacy.cheap/# online pharmacy discount code
cheap pharmacy no prescription: canada pharmacy online – canadian pharmacy world coupon
I’m always excited to see your posts in my feed. Another excellent article!
Wow, amazing weblog layout! How long have you ever been blogging for?
you made running a blog glance easy. The entire look of your web site
is magnificent, let alone the content! You can see similar here dobry sklep
best online pharmacy without prescription: canada pharmacies online prescriptions – how to order prescription drugs from canada
no prescription medicines no prescription pharmacy online order prescription drugs online without doctor
purchasing prescription drugs online: online meds without prescription – canadian pharmacy no prescription
https://onlinepharmacy.cheap/# best no prescription pharmacy
https://mexicanpharm.online/# mexico drug stores pharmacies
best no prescription online pharmacy: best online pharmacy no prescription – buy medication online no prescription
acillin sale cheap penicillin cheap amoxil generic
http://mexicanpharm.online/# buying from online mexican pharmacy
best online canadian pharmacy: canadian pharmacies compare – buy canadian drugs
india pharmacy: indian pharmacies safe – best india pharmacy
furosemide 100mg generic – candesartan 16mg drug buy capoten without a prescription
online pharmacies without prescriptions buying prescription drugs online canada buy drugs online without a prescription
https://indianpharm.shop/# india pharmacy mail order
http://canadianpharm.guru/# canadian pharmacy online reviews
online medication no prescription: best website to buy prescription drugs – buying prescription medicine online
I’m impressed by your ability to convey such nuanced ideas with clarity.
indian pharmacy online: indian pharmacy paypal – indian pharmacy paypal
Online medicine home delivery: mail order pharmacy india – Online medicine order
https://pharmacynoprescription.pro/# buy prescription drugs online without doctor
canadian pharmacy mall: best online canadian pharmacy – canadian pharmacy world
indianpharmacy com: п»їlegitimate online pharmacies india – pharmacy website india
online pharmacy india: buy medicines online in india – Online medicine home delivery
http://canadianpharm.guru/# canadian drug prices
canada pharmacy reviews pharmacy wholesalers canada thecanadianpharmacy
reputable mexican pharmacies online: mexican pharmaceuticals online – mexican online pharmacies prescription drugs
I’m so grateful for the information you’ve shared. It’s been incredibly enlightening!
mexican mail order pharmacies: mexican pharmaceuticals online – mexico pharmacy
my canadian pharmacy review: my canadian pharmacy review – canadian drugs pharmacy
Such a well-researched piece! It’s evident how much effort you’ve put in.
This post was a breath of fresh air. Thank you for your unique insights!
https://canadianpharm.guru/# is canadian pharmacy legit
canadian drug: canadian pharmacy antibiotics – maple leaf pharmacy in canada
I always learn something new from your posts. Thank you for the education!
I’m bookmarking this for future reference. Your advice is spot on!
canadian pharmacy drugs online: pharmacy canadian – canada rx pharmacy world
purple pharmacy mexico price list: mexico drug stores pharmacies – mexico drug stores pharmacies
mexican border pharmacies shipping to usa: medicine in mexico pharmacies – mexico pharmacies prescription drugs
https://mexicanpharm.online/# mexico drug stores pharmacies
Your post is a ray of light in the darkness. Thank you for brightening my day in a unique way. Keep shining!
Your insights have added a lot of value to my understanding. Thanks for sharing.
canadian online pharmacy reviews: cheap canadian pharmacy online – safe reliable canadian pharmacy
I have no words to describe how your content illuminated my day. Keep being that source of inspiration!
http://indianpharm.shop/# indian pharmacy
medication from mexico pharmacy mexico pharmacies prescription drugs reputable mexican pharmacies online
https://pharmacynoprescription.pro/# buy medications without prescriptions
canadian pharmacy online ship to usa: pharmacy canadian superstore – reliable canadian pharmacy
canada mail order prescriptions: prescription drugs online canada – canada drugs no prescription
remarkable day starting with a phenomenal reading
cheapest online pharmacy india: best india pharmacy – indian pharmacy paypal
mexican pharmacy: mexican online pharmacies prescription drugs – mexican border pharmacies shipping to usa
https://indianpharm.shop/# india pharmacy mail order
Your post resonated with me on many levels. Thank you for writing it!
mexican border pharmacies shipping to usa: mexican online pharmacies prescription drugs – mexico drug stores pharmacies
indian pharmacy: buy medicines online in india – indian pharmacy online
Your blog is a constant source of inspiration and knowledge. Thank you!
https://indianpharm.shop/# Online medicine order
buy prescription drugs without a prescription: canadian prescription – discount prescription drugs canada
canadian prescription: legitimate online pharmacy no prescription – non prescription canadian pharmacy
canadian pharmacy sarasota: canadianpharmacymeds com – canadian online pharmacy
https://indianpharm.shop/# cheapest online pharmacy india
safe canadian pharmacy canadianpharmacy com online canadian pharmacy reviews
canadian pharmacy without a prescription: canadian prescription – non prescription canadian pharmacy
https://pharmacynoprescription.pro/# canadian and international prescription service
mexican rx online: mexican rx online – best online pharmacies in mexico
top 10 online pharmacy in india: indian pharmacy – п»їlegitimate online pharmacies india
quality prescription drugs canada: non prescription online pharmacy – mexican pharmacies no prescription
mexican pharmacy: buying from online mexican pharmacy – mexico drug stores pharmacies
http://indianpharm.shop/# indian pharmacy online
canadian pharmacy prices: cross border pharmacy canada – safe online pharmacies in canada
medicine with no prescription: meds no prescription – canadian prescription drugstore reviews
https://pharmacynoprescription.pro/# best no prescription online pharmacy
buying prescription drugs in mexico online: buying prescription drugs in mexico online – mexican drugstore online
http://pharmacynoprescription.pro/# online no prescription pharmacy
canadian pharmacy online no prescription needed buy medication online without prescription mail order prescriptions from canada
retrovir for sale online – order biaxsig cheap zyloprim 300mg
fabulous sunrise initiating with a wonderful literature
india online pharmacy: best india pharmacy – reputable indian online pharmacy
purchase glycomet pill – glycomet 1000mg tablet buy generic lincomycin
indian pharmacy: indian pharmacies safe – indianpharmacy com
https://indianpharm.shop/# online pharmacy india
best india pharmacy: mail order pharmacy india – buy medicines online in india
best canadian pharmacy online: best canadian pharmacy to order from – is canadian pharmacy legit
https://mexicanpharm.online/# buying from online mexican pharmacy
reputable indian pharmacies: buy medicines online in india – legitimate online pharmacies india
real canadian pharmacy: drugs from canada – canadian pharmacy tampa
online no prescription pharmacy: no prescription pharmacy – buying prescription drugs from canada online
canadian pharmacy no scripts: best canadian online pharmacy – legit canadian pharmacy
no prescription medication ordering prescription drugs from canada canada online prescription
Your attention to detail is remarkable. I appreciate the thoroughness of your post.
http://canadianpharm.guru/# reliable canadian pharmacy reviews
buy medications without a prescription: buy medications online without prescription – pharmacy no prescription required
https://slotsiteleri.guru/# casino slot siteleri
http://pinupgiris.fun/# pin up giris
http://gatesofolympus.auction/# gates of olympus giris
pin up bet: pin up aviator – pin-up casino giris
https://gatesofolympus.auction/# gates of olympus oyna demo
aviator sinyal hilesi ucretsiz: aviator sinyal hilesi ucretsiz – aviator nas?l oynan?r
https://slotsiteleri.guru/# güvenilir slot siteleri 2024
This post was a breath of fresh air. Thank you for your unique insights!
sweet bonanza yasal site: sweet bonanza giris – guncel sweet bonanza
http://slotsiteleri.guru/# güvenilir slot siteleri
http://slotsiteleri.guru/# slot bahis siteleri
pin up: pin up bet – pin up bet
http://aviatoroyna.bid/# aviator oyna
https://slotsiteleri.guru/# 2024 en iyi slot siteleri
ucak oyunu bahis aviator: aviator nas?l oynan?r – aviator sinyal hilesi apk
aviator hile: aviator oyunu giris – ucak oyunu bahis aviator
https://aviatoroyna.bid/# aviator bahis
aviator sinyal hilesi ucretsiz: aviator mostbet – aviator hilesi
http://sweetbonanza.bid/# sweet bonanza taktik
https://slotsiteleri.guru/# güvenilir slot siteleri
http://aviatoroyna.bid/# aviator oyna slot
purchase quetiapine pill – trazodone pills purchase eskalith
sweet bonanza yasal site: sweet bonanza demo – sweet bonanza
https://aviatoroyna.bid/# aviator casino oyunu
aviator oyna 20 tl: aviator bahis – ucak oyunu bahis aviator
pin-up casino giris: pin up bet – pin-up online
http://pinupgiris.fun/# pin up casino giris
http://pinupgiris.fun/# pin-up online
pin-up giris: pin-up online – pin-up online
https://sweetbonanza.bid/# sweet bonanza nasil oynanir
ucak oyunu bahis aviator: aviator sinyal hilesi apk – aviator sinyal hilesi
sweet bonanza bahis: sweet bonanza free spin demo – sweet bonanza bahis
http://gatesofolympus.auction/# gates of olympus oyna
http://gatesofolympus.auction/# gates of olympus taktik
yasal slot siteleri: deneme bonusu veren siteler – en guvenilir slot siteleri
buying prescription drugs in mexico: mexico pharmacies prescription drugs – medication from mexico pharmacy
purple pharmacy mexico price list Online Pharmacies in Mexico reputable mexican pharmacies online
mexican pharmacy: mexico pharmacy – п»їbest mexican online pharmacies
mexican online pharmacies prescription drugs mexican pharmacy mexico pharmacy
https://indianpharmacy.icu/# india online pharmacy
reputable indian online pharmacy indian pharmacy delivery mail order pharmacy india
buying prescription drugs in mexico: Mexican Pharmacy Online – mexican online pharmacies prescription drugs
india pharmacy mail order: Healthcare and medicines from India – buy prescription drugs from india
Your insights have added a lot of value to my understanding. Thanks for sharing.
atarax sale – buy buspin buy amitriptyline sale
canada pharmacy world: Certified Canadian Pharmacy – canadian world pharmacy
buy prescription drugs from india Healthcare and medicines from India top 10 pharmacies in india
buying from online mexican pharmacy: cheapest mexico drugs – medication from mexico pharmacy
order clomipramine 50mg generic – order abilify 30mg online buy sinequan pills
canada drug pharmacy: northwest pharmacy canada – recommended canadian pharmacies
canadian drug stores canadian online drugstore best canadian online pharmacy reviews
What a compelling enjoy reading! Your arguments were well-presented and convincing.
canadian drug: canadian pharmacy 24 – vipps approved canadian online pharmacy
legit canadian pharmacy online: ed meds online canada – canadian pharmacies that deliver to the us
canadian pharmacy mall: canadian pharmacy 24 – pharmacy com canada
pharmacy in canada Large Selection of Medications canada ed drugs
mexican pharmacy: Mexican Pharmacy Online – mexico drug stores pharmacies
pharmacies in mexico that ship to usa: Online Pharmacies in Mexico – reputable mexican pharmacies online
canadian mail order pharmacy Large Selection of Medications canadian neighbor pharmacy
Online medicine order indian pharmacy reputable indian pharmacies
mexico pharmacies prescription drugs: Mexican Pharmacy Online – mexico pharmacy
indianpharmacy com: indian pharmacy – buy medicines online in india
cheap canadian pharmacy Licensed Canadian Pharmacy onlinecanadianpharmacy 24
india pharmacy: indian pharmacies safe – top 10 online pharmacy in india
world pharmacy india: indian pharmacy – online shopping pharmacy india
reputable indian pharmacies Healthcare and medicines from India indian pharmacy paypal
mexican pharmacy: cheapest mexico drugs – mexican online pharmacies prescription drugs
https://zithromaxall.shop/# can i buy zithromax online
how to get amoxicillin how to buy amoxicillin online generic amoxicillin 500mg
https://clomidall.com/# can you get generic clomid tablets
http://prednisoneall.shop/# prednisone uk buy
amoxicillin cost australia: price of amoxicillin without insurance – amoxicillin 30 capsules price
https://prednisoneall.com/# prednisone without prescription 10mg
amoxicillin tablets in india buying amoxicillin online ampicillin amoxicillin
prednisone 20 mg prices: prednisone otc uk – prednisone pills cost
https://prednisoneall.shop/# prednisone 20mg prescription cost
I’m always excited to see your posts in my feed. Another excellent article!
This article was a joy to enjoy reading. Your enthusiasm is contagious!
rx clomid order generic clomid tablets where to get generic clomid online
https://prednisoneall.com/# where can i buy prednisone
https://prednisoneall.com/# prednisone 30 mg tablet
can you buy generic clomid without insurance buy clomid no prescription where to get clomid without insurance
https://clomidall.shop/# where can i get cheap clomid
amoxicillin drug – cefadroxil 500mg tablet cipro oral
https://prednisoneall.com/# prednisone in uk
Neon Flying Squid
https://www.gyogyteabolt.hu/component/k2/item/136-altalanos-szerzodesi-felteteleklimit=10?limit=10
I’m so glad I stumbled upon this article. It was exactly what I needed to enjoy reading!
can i get clomid no prescription buy generic clomid without insurance how to buy generic clomid
http://clomidall.shop/# order cheap clomid without prescription
zithromax: buy zithromax no prescription – buy zithromax online
http://amoxilall.shop/# can you buy amoxicillin over the counter
http://amoxilall.shop/# buy amoxicillin online mexico
where can i get zithromax generic zithromax india buy zithromax online
http://zithromaxall.com/# zithromax over the counter uk
https://amoxilall.com/# where can i buy amoxicillin over the counter
order augmentin 375mg online – buy augmentin 1000mg pills cipro for sale online
amoxicillin 500 mg over the counter amoxicillin canada amoxicillin order online no prescription
Neon Flying Squid
https://www.parcheggiopinguino.it/cooking-lessons-with-our-chef/
Neon Flying Squid
http://fisioterapiaalc.es/index.php/ca/our-doctors/item/7-accumsan-lobortis-metus-mauris-at-nibh?start=8050
http://prednisoneall.com/# prednisone rx coupon
http://amoxilall.shop/# buy cheap amoxicillin online
price for amoxicillin 875 mg can i buy amoxicillin over the counter amoxicillin 500 capsule
https://zithromaxall.com/# where can i get zithromax
prednisone coupon: prednisone 54 – prednisone 5mg cost
https://amoxilall.shop/# amoxicillin 500 mg tablet price
http://prednisoneall.shop/# prednisone 40mg
zithromax 500mg zithromax online paypal buy zithromax online with mastercard
Cheap Cialis: cheapest cialis – Buy Cialis online
https://kamagraiq.com/# sildenafil oral jelly 100mg kamagra
Generic Viagra online Order Viagra 50 mg online п»їBuy generic 100mg Viagra online
buy Viagra online: best price on viagra – sildenafil online
https://kamagraiq.com/# Kamagra tablets
п»їBuy generic 100mg Viagra online viagra canada sildenafil 50 mg price
Viagra tablet online: best price for viagra 100mg – sildenafil online
Kamagra 100mg price: kamagra best price – buy kamagra online usa
cheapest cialis: Generic Tadalafil 20mg price – Generic Cialis without a doctor prescription
п»їkamagra Kamagra Iq cheap kamagra
http://tadalafiliq.shop/# Cialis without a doctor prescription
https://tadalafiliq.com/# Buy Tadalafil 20mg
Viagra online price: Viagra online price – Cheap Sildenafil 100mg
Kamagra tablets Kamagra Iq Kamagra Oral Jelly
Generic Viagra online: generic ed pills – generic sildenafil
Cialis over the counter: cheapest cialis – Cialis 20mg price
http://sildenafiliq.xyz/# buy Viagra online
Generic Viagra for sale: cheapest viagra – Generic Viagra for sale
buy Kamagra Kamagra Iq buy Kamagra
viagra without prescription: best price on viagra – cheapest viagra
http://kamagraiq.com/# Kamagra Oral Jelly
https://tadalafiliq.com/# Cialis 20mg price
Viagra generic over the counter cheapest viagra Cheap Viagra 100mg
buy Viagra over the counter: viagra without prescription – generic sildenafil
buy Viagra over the counter: generic ed pills – Cheap Sildenafil 100mg
Kamagra 100mg price kamagra best price Kamagra 100mg price
Kamagra 100mg price: Kamagra gel – buy Kamagra
https://tadalafiliq.shop/# cheapest cialis
Cheap generic Viagra online: best price on viagra – sildenafil 50 mg price
cheap kamagra Kamagra Oral Jelly Price sildenafil oral jelly 100mg kamagra
https://kamagraiq.shop/# super kamagra
buy kamagra online usa: Kamagra gel – sildenafil oral jelly 100mg kamagra
http://kamagraiq.com/# Kamagra tablets
Hi, always i used to check webpage posts here early in the dawn, for the reason that i love to learn more and
more.
buy Kamagra: Kamagra gel – kamagra
buy kamagra online usa Kamagra Iq Kamagra 100mg price
https://kamagraiq.shop/# Kamagra tablets
viagra canada: over the counter sildenafil – sildenafil online
http://tadalafiliq.shop/# п»їcialis generic
buy Kamagra Kamagra Oral Jelly Price Kamagra 100mg
Cialis 20mg price in USA: Buy Cialis online – Tadalafil price
https://kamagraiq.shop/# Kamagra 100mg
Cheap Viagra 100mg: generic ed pills – order viagra
https://kamagraiq.com/# cheap kamagra
http://canadianpharmgrx.xyz/# canadian pharmacy 365
india pharmacy mail order Healthcare and medicines from India Online medicine home delivery
What’s up to every one, the contents present at this site
are actually amazing for people experience, well, keep up
the nice work fellows.
https://indianpharmgrx.shop/# indianpharmacy com
canadian pharmacy 24 com List of Canadian pharmacies online canadian pharmacy
maple leaf pharmacy in canada: CIPA approved pharmacies – canada ed drugs
https://canadianpharmgrx.xyz/# canadianpharmacy com
I am regular reader, how are you everybody? This piece of writing
posted at this website is genuinely fastidious.
reputable indian pharmacies Healthcare and medicines from India Online medicine home delivery
https://mexicanpharmgrx.com/# mexican online pharmacies prescription drugs
order cleocin 300mg online cheap – order cleocin 300mg online cheap chloromycetin us
https://mexicanpharmgrx.com/# mexico pharmacy
buy medicines online in india Healthcare and medicines from India india online pharmacy
https://mexicanpharmgrx.com/# mexico pharmacies prescription drugs
Hi it’s me, I am also visiting this site daily, this web page is in fact good and the
visitors are really sharing good thoughts.
mexican pharmacy online pharmacy in Mexico mexico pharmacies prescription drugs
onlinepharmaciescanada com: Cheapest drug prices Canada – canada drug pharmacy
https://canadianpharmgrx.xyz/# the canadian pharmacy
indian pharmacies safe Generic Medicine India to USA top 10 pharmacies in india
What is a Sugar Defender? Sugar Shield could be an affront affectability enhancement product that effectively supports stable blood sugar levels.
cheapest online pharmacy india: indian pharmacy – top online pharmacy india
https://canadianpharmgrx.xyz/# onlinepharmaciescanada com
buy medicines online in india Generic Medicine India to USA mail order pharmacy india
https://mexicanpharmgrx.com/# mexican pharmaceuticals online
https://indianpharmgrx.shop/# indian pharmacies safe
I appreciate the balance and fairness in your writing. Great job!
mexico pharmacies prescription drugs mexican pharmacy mexican online pharmacies prescription drugs
mexico drug stores pharmacies: online pharmacy in Mexico – mexican rx online
https://canadianpharmgrx.xyz/# canadian online drugstore
pharmacy website india: Generic Medicine India to USA – india pharmacy
azithromycin order – buy flagyl pills for sale buy ciplox generic
http://mexicanpharmgrx.shop/# buying prescription drugs in mexico online
best online pharmacies in mexico online pharmacy in Mexico mexico drug stores pharmacies
http://indianpharmgrx.shop/# best india pharmacy
buying prescription drugs in mexico online mexican pharmacy pharmacies in mexico that ship to usa
http://mexicanpharmgrx.shop/# buying from online mexican pharmacy
https://canadianpharmgrx.com/# canadian online pharmacy
buy cipro online without prescription: cipro – ciprofloxacin 500 mg tablet price
pct nolvadex: tamoxifen and bone density – natural alternatives to tamoxifen
buy cipro online cipro online no prescription in the usa purchase cipro
This design is incredible! You obviously know how to keep a reader entertained. Between your wit and your videos, I was almost moved to start my own blog (well, almost…HaHa!) Fantastic job. I really enjoyed what you had to say, and more than that, how you presented it. Too cool!
I’m always excited to see your posts in my feed. Another excellent article!
I’m in awe of the way you handle topics with both grace and authority.
100mg doxycycline: online doxycycline – doxycycline medication
Cytotec 200mcg price Cytotec 200mcg price buy cytotec online
nolvadex estrogen blocker: tamoxifen alternatives – tamoxifen hair loss
tamoxifen mechanism of action: tamoxifen postmenopausal – nolvadex estrogen blocker
buy cytotec pills online cheap order cytotec online buy cytotec online
tamoxifen: tamoxifen vs raloxifene – tamoxifen rash
aromatase inhibitors tamoxifen: does tamoxifen cause weight loss – is nolvadex legal
doxycycline mono order doxycycline buy doxycycline monohydrate
tamoxifen men: tamoxifen 20 mg tablet – tamoxifen pill
tamoxifen warning lexapro and tamoxifen nolvadex pills
buy diflucan: diflucan 1 where to buy – diflucan tablets online
buy cytotec online Abortion pills online buy cytotec online fast delivery
aromatase inhibitor tamoxifen: tamoxifenworld – nolvadex 10mg
albuterol 2mg inhaler – fluticasone brand order theo-24 Cr 400mg for sale
https://nolvadex.icu/# who should take tamoxifen
order doxycycline 100mg without prescription: buy generic doxycycline – purchase doxycycline online
Cytotec 200mcg price Cytotec 200mcg price cytotec buy online usa
The writing captivated me from the first paragraph to the last. It’s rare to find such engaging content.
buy generic ciprofloxacin: ciprofloxacin order online – buy generic ciprofloxacin
order cytotec online Abortion pills online purchase cytotec
ciprofloxacin 500mg buy online: cipro 500mg best prices – ciprofloxacin
ivermectin 6mg pills – buy levaquin 500mg generic order cefaclor 250mg online cheap
cytotec online: cytotec buy online usa – order cytotec online
doxycycline 100mg online doxycycline hyclate 100 mg cap doxycycline 200 mg
http://nolvadex.icu/# tamoxifen vs raloxifene
can you buy diflucan otc: diflucan 50 mg capsule – diflucan generic costs
buy cipro cheap: п»їcipro generic – cipro online no prescription in the usa
buy diflucan over the counter diflucan 50 mg capsule can you buy diflucan over the counter uk
price of doxycycline: buy generic doxycycline – doxycycline 100mg capsules
cipro pharmacy: buy generic ciprofloxacin – cipro pharmacy
buy cytotec Misoprostol 200 mg buy online buy cytotec over the counter
diflucan capsule 50 mg: buy diflucan without prescription – how to get diflucan over the counter
prednisone 10 mg brand name: how can i get prednisone online without a prescription – prednisone 10mg price in india
http://azithromycina.pro/# order zithromax without prescription
how to get zithromax zithromax without prescription zithromax online australia
can i buy zithromax online: generic zithromax medicine – buy cheap zithromax online
3000mg prednisone prednisone 10mg cost prednisone buy canada
buy generic zithromax online: zithromax z-pak – zithromax capsules
how much is zithromax 250 mg: zithromax online usa no prescription – buy generic zithromax online
This was a great enjoy reading—thought-provoking and informative. Thank you!
I always learn something new from your posts. Thank you for the education!
https://amoxicillina.top/# amoxicillin 500mg cost
amoxicillin 875 125 mg tab amoxicillin 500mg capsules uk amoxicillin 500 mg without prescription
zithromax 500 mg lowest price pharmacy online: zithromax online – zithromax pill
https://prednisonea.store/# prednisone 10mg prices
order desloratadine 5mg pills – oral desloratadine albuterol cheap
amoxicillin over the counter in canada: amoxicillin where to get – amoxicillin 500 mg
buy generic clomid no prescription: how to buy cheap clomid price – can i get clomid no prescription
prednisone 4mg how to get prednisone tablets prednisone 4 mg daily
Your work is truly inspirational. I appreciate the depth you bring to your topics.
This post is a testament to your expertise and hard work. Thank you!
prednisone 2 5 mg: prednisone 10 mg coupon – prednisone pharmacy
https://azithromycina.pro/# zithromax capsules australia
ivermectin lotion 0.5 stromectol prices stromectol liquid
order generic clomid: can i buy generic clomid no prescription – can you get cheap clomid price
http://clomida.pro/# can you get generic clomid prices
zithromax 250 mg: zithromax 250 mg tablet price – where can i buy zithromax capsules
zithromax online usa: zithromax antibiotic without prescription – can i buy zithromax over the counter in canada
canada pharmacy not requiring prescription pharmacy without prescription canadian pharmacy coupon
where to buy erectile dysfunction pills: cost of ed meds – erectile dysfunction medicine online
http://edpill.top/# cheap boner pills
Wow, wonderful weblog structure! How lengthy have you ever been running a
blog for? you make blogging glance easy. The entire
glance of your website is wonderful, as smartly as the
content material! You can see similar here dobry sklep
Just what I was looking for, appreciate it for putting up.
https://medicationnoprescription.pro/# buying prescription drugs in india
http://medicationnoprescription.pro/# buying prescription drugs in canada
canadian pharmacy coupon prescription free canadian pharmacy canadian pharmacy coupon
pharmacy without prescription: canadian pharmacy coupon code – no prescription pharmacy paypal
What i don’t realize is if truth be told how you’re no longer actually a lot more well-favored than you might be now. You are so intelligent. You recognize therefore considerably on the subject of this matter, made me in my view imagine it from numerous various angles. Its like women and men don’t seem to be interested unless it is something to accomplish with Girl gaga! Your personal stuffs excellent. All the time take care of it up!
https://edpill.top/# buy ed meds online
Great amazing things here. I?¦m very satisfied to see your article. Thank you a lot and i’m taking a look ahead to contact you. Will you kindly drop me a mail?
methylprednisolone tablet – fml-forte sale cost azelastine
cheapest pharmacy to fill prescriptions without insurance: pharmacy coupons – non prescription medicine pharmacy
meds no prescription buy meds online no prescription medicine with no prescription
online medication no prescription: prescription from canada – online pharmacies without prescriptions
https://onlinepharmacyworld.shop/# canadian prescription pharmacy
http://edpill.top/# buy ed medication
canadian and international prescription service: no prescription online pharmacies – no prescription needed
online erectile dysfunction prescription top rated ed pills cheap ed drugs
https://onlinepharmacyworld.shop/# pharmacy coupons
no prescription online pharmacy: can you buy prescription drugs in canada – no prescription canadian pharmacy
https://medicationnoprescription.pro/# order medication without prescription
how to get ed meds online discount ed pills boner pills online
https://onlinepharmacyworld.shop/# prescription drugs online
buy micronase 2.5mg generic – generic glipizide 10mg forxiga pills
where to get ed pills: low cost ed meds – buying ed pills online
https://casinvietnam.shop/# web c? b?c online uy tin
dánh bài tr?c tuy?n: casino tr?c tuy?n vi?t nam – casino tr?c tuy?n
https://casinvietnam.com/# casino tr?c tuy?n uy tin
web c? b?c online uy tin web c? b?c online uy tin casino tr?c tuy?n vi?t nam
game c? b?c online uy tín: choi casino tr?c tuy?n trên di?n tho?i – casino tr?c tuy?n uy tín
casino tr?c tuy?n uy tin casino tr?c tuy?n vi?t nam casino tr?c tuy?n uy tin
http://casinvietnam.com/# casino tr?c tuy?n vi?t nam
отмечу как доверительного союзника в SEO-развитии.
This paragraph will help the internet users for building up new
website or even a weblog from start to end.
Обращение в seo-prodvijenie-saytov.ru для поисковой оптимизации моего сайта, когда заметил снижение трафика. Результаты были быстро заметны: компетентность команды, отзывчивые специалисты и видимый рост позиций. Рекомендую!
PBN sites
We’ll create a system of self-owned blog network sites!
Advantages of our PBN network:
WE DO everything SO THAT GOOGLE does not understand that this A self-owned blog network!!!
1- We buy domain names from distinct registrars
2- The principal site is hosted on a VPS server (Virtual Private Server is high-speed hosting)
3- Additional sites are on various hostings
4- We allocate a individual Google account to each site with verification in Google Search Console.
5- We design websites on WordPress, we don’t employ plugins with aided by which Trojans penetrate and through which pages on your websites are generated.
6- We refrain from reproduce templates and utilize only unique text and pictures
We refrain from work with website design; the client, if wanted, can then edit the websites to suit his wishes
casino tr?c tuy?n vi?t nam: casino tr?c tuy?n uy tín – casino online uy tín
casino tr?c tuy?n: choi casino tr?c tuy?n trên di?n tho?i – casino tr?c tuy?n uy tín
This article was a joy to enjoy reading. Your enthusiasm is contagious!
http://casinvietnam.shop/# casino tr?c tuy?n
choi casino tr?c tuy?n trên di?n tho?i: casino online uy tín – casino tr?c tuy?n
http://casinvietnam.shop/# danh bai tr?c tuy?n
casino tr?c tuy?n vi?t nam: game c? b?c online uy tín – casino tr?c tuy?n uy tín
prandin tablet – empagliflozin for sale online order jardiance 25mg online
casino tr?c tuy?n: casino tr?c tuy?n vi?t nam – game c? b?c online uy tín
http://casinvietnam.com/# danh bai tr?c tuy?n
Thanks – Enjoyed this blog post, is there any way I can receive an email whenever you publish a fresh article?
casino tr?c tuy?n vi?t nam: web c? b?c online uy tín – web c? b?c online uy tín
Puravive is a weight loss supplement that targets the root cause of weight gain issues in men and women.
http://casinvietnam.com/# casino tr?c tuy?n uy tin
casino tr?c tuy?n: casino tr?c tuy?n – casino tr?c tuy?n uy tín
web c? b?c online uy tin game c? b?c online uy tin casino tr?c tuy?n
https://casinvietnam.com/# casino tr?c tuy?n
casino tr?c tuy?n uy tín: casino tr?c tuy?n uy tín – casino online uy tín
casino online uy tin casino tr?c tuy?n uy tin casino tr?c tuy?n vi?t nam
Your attention to detail is remarkable. I appreciate the thoroughness of your post.
game c? b?c online uy tín: dánh bài tr?c tuy?n – casino online uy tín
https://casinvietnam.com/# casino tr?c tuy?n vi?t nam
casino tr?c tuy?n choi casino tr?c tuy?n tren di?n tho?i casino online uy tin
choi casino tr?c tuy?n trên di?n tho?i: casino tr?c tuy?n vi?t nam – casino online uy tín
http://casinvietnam.shop/# casino tr?c tuy?n vi?t nam
What Is Exactly ZenCortex? ZenCortex is an optimal hearing function support
choi casino tr?c tuy?n tren di?n tho?i casino tr?c tuy?n vi?t nam casino tr?c tuy?n uy tin
choi casino tr?c tuy?n trên di?n tho?i: game c? b?c online uy tín – casino online uy tín
http://casinvietnam.shop/# game c? b?c online uy tin
casino tr?c tuy?n vi?t nam: choi casino tr?c tuy?n trên di?n tho?i – web c? b?c online uy tín
http://casinvietnam.com/# choi casino tr?c tuy?n tren di?n tho?i
casino tr?c tuy?n vi?t nam: casino tr?c tuy?n vi?t nam – casino tr?c tuy?n
web c? b?c online uy tin game c? b?c online uy tin casino tr?c tuy?n vi?t nam
casino tr?c tuy?n: casino tr?c tuy?n uy tín – casino tr?c tuy?n vi?t nam
casino tr?c tuy?n casino tr?c tuy?n vi?t nam casino tr?c tuy?n vi?t nam
http://indiaph24.store/# top 10 online pharmacy in india
top 10 pharmacies in india Cheapest online pharmacy top 10 online pharmacy in india
http://canadaph24.pro/# cheapest pharmacy canada
mexican mail order pharmacies: Mexican Pharmacy Online – medicine in mexico pharmacies
https://canadaph24.pro/# cross border pharmacy canada
order glucophage 500mg generic – buy januvia 100 mg generic buy precose
mexican pharmaceuticals online Mexican Pharmacy Online medicine in mexico pharmacies
pharmacy website india https://indiaph24.store/# online pharmacy india
india pharmacy
https://mexicoph24.life/# mexico drug stores pharmacies
best canadian pharmacy Certified Canadian Pharmacies online canadian pharmacy
https://canadaph24.pro/# best canadian pharmacy to buy from
mexican rx online best online pharmacies in mexico pharmacies in mexico that ship to usa
https://canadaph24.pro/# canada drug pharmacy
http://indiaph24.store/# indian pharmacy
cheapest online pharmacy india indian pharmacy fast delivery Online medicine order
mexican rx online: mexico pharmacy – mexican mail order pharmacies
https://canadaph24.pro/# canadian pharmacy 365
mexico pharmacy Online Pharmacies in Mexico mexican mail order pharmacies
http://indiaph24.store/# online shopping pharmacy india
top online pharmacy india buy medicines from India india online pharmacy
http://mexicoph24.life/# buying from online mexican pharmacy
reputable canadian online pharmacies canadian online pharmacy reviews legit canadian pharmacy
https://indiaph24.store/# reputable indian online pharmacy
canadian drug canadian pharmacies canadian pharmacy com
http://mexicoph24.life/# п»їbest mexican online pharmacies
http://canadaph24.pro/# pharmacy in canada
buy medicines online in india indian pharmacy best india pharmacy
vipps approved canadian online pharmacy: Prescription Drugs from Canada – legitimate canadian online pharmacies
http://indiaph24.store/# indian pharmacies safe
mexico pharmacies prescription drugs Mexican Pharmacy Online mexico drug stores pharmacies
http://canadaph24.pro/# best canadian pharmacy to buy from
canadian pharmacy phone number Certified Canadian Pharmacies canadian drug pharmacy
online pharmacy india: top 10 pharmacies in india – indian pharmacies safe
purchase terbinafine generic – order fulvicin online cheap buy griseofulvin pills
This post is packed with useful insights. Thanks for sharing your knowledge!
https://indiaph24.store/# best india pharmacy
cheapest online pharmacy india Cheapest online pharmacy best india pharmacy
Online medicine home delivery indian pharmacy online cheapest online pharmacy india
https://indiaph24.store/# legitimate online pharmacies india
northwest pharmacy canada canada pharmacy world reputable canadian online pharmacies
https://canadaph24.pro/# canadian online pharmacy reviews
reliable canadian pharmacy: Prescription Drugs from Canada – canadian 24 hour pharmacy
https://canadaph24.pro/# canada discount pharmacy
п»їlegitimate online pharmacies india indian pharmacy buy prescription drugs from india
http://indiaph24.store/# indian pharmacy
reputable indian pharmacies indian pharmacy fast delivery top 10 pharmacies in india
https://indiaph24.store/# best india pharmacy
https://mexicoph24.life/# mexican pharmaceuticals online
mexican border pharmacies shipping to usa Mexican Pharmacy Online mexico pharmacy
Quality content is the main to be a focus for the visitors to go
to see the website, that’s what this website is providing.
https://mexicoph24.life/# mexico drug stores pharmacies
canadian pharmacy price checker Certified Canadian Pharmacies canadian pharmacy checker
Your ability to distill complex concepts into enjoy readingable content is admirable.
https://mexicoph24.life/# medication from mexico pharmacy
purple pharmacy mexico price list mexican drugstore online mexican pharmaceuticals online
This is one of the most comprehensive articles I’ve enjoy reading on this topic. Kudos!
https://canadaph24.pro/# canada pharmacy
canadian pharmacy 24 best canadian pharmacy online canadian pharmacy online ship to usa
http://mexicoph24.life/# best mexican online pharmacies
mexico pharmacy mexico pharmacy buying prescription drugs in mexico online
http://indiaph24.store/# world pharmacy india
top online pharmacy india indian pharmacy best online pharmacy india
https://mexicoph24.life/# mexican drugstore online
The very root of your writing whilst sounding reasonable in the beginning, did not sit well with me after some time. Someplace throughout the paragraphs you were able to make me a believer unfortunately only for a while. I nevertheless have got a problem with your leaps in assumptions and one might do nicely to fill in those breaks. If you can accomplish that, I will surely end up being fascinated.
canadian pharmacy uk delivery Licensed Canadian Pharmacy canadian pharmacy india
http://canadaph24.pro/# canadian mail order pharmacy
india pharmacy Generic Medicine India to USA indian pharmacies safe
Such a well-researched piece! It’s evident how much effort you’ve put in.
buy semaglutide 14mg – rybelsus 14mg without prescription DDAVP medication
https://indiaph24.store/# top 10 pharmacies in india
medication from mexico pharmacy mexico pharmacy buying prescription drugs in mexico online
https://mexicoph24.life/# medicine in mexico pharmacies
mexican drugstore online mexico drug stores pharmacies medication from mexico pharmacy
https://canadaph24.pro/# canadian pharmacy 24 com
online canadian pharmacy canadian pharmacies canadian pharmacy king
https://indiaph24.store/# india pharmacy mail order
purple pharmacy mexico price list mexican pharmacy mexican pharmaceuticals online
http://canadaph24.pro/# canadian pharmacy mall
buy nizoral 200mg generic – cheap nizoral sporanox drug
http://mexicoph24.life/# purple pharmacy mexico price list
online shopping pharmacy india indian pharmacy fast delivery best india pharmacy
http://indiaph24.store/# indianpharmacy com
buy prescription drugs from india Cheapest online pharmacy top online pharmacy india
https://mexicoph24.life/# mexican rx online
canadian pharmacy 1 internet online drugstore Licensed Canadian Pharmacy legit canadian pharmacy
http://indiaph24.store/# indian pharmacy